-
May 15, 2026 Guy Sauvageau Honored by the Stem Cell Network
Professor Guy Sauvageau, director of the Molecular Genetics of Stem Cells Research Unit, has been awarded the Stem Cell Network’s Trailblazer Award. This award recognizes individuals who have overcome major challenges in the field of regenerative medicine and developed solutions that advance care, policy, commercialization, or innovation within a system.
![]()
-
May 6, 2026 Guy Sauvageau Named Knight of the Ordre de Montréal
On May 8, Professor Guy Sauvageau, Director of IRIC’s Molecular Genetics of Stem Cells Research Unit, will be presented with the insignia of the Ordre de Montréal as a Knight.
![]()
-
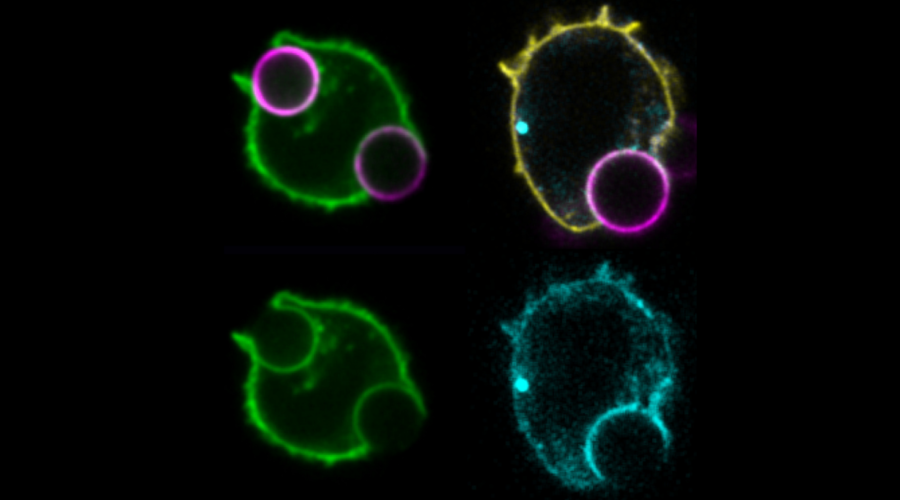
April 30, 2026 RIMap-RISC: A New Interface to Study RNA Biology
The team led by François Major, director of IRIC’s RNA Engineering Research Unit, has developed the new RIMap-RISC database, which integrates the molecular structure of microRNAs and messenger RNAs to systematically model their interactions. Led by PhD student Simon Chasles, this work is the subject of a publication in Genome Biology.
![]()
-
April 8, 2026 Marc Therrien’s team wins the 2025 Discovery of the Year Award - Québec Science
The team led by Marc Therrien, Principal Investigator and Chief Executive Officer at IRIC, has won Québec Science magazine’s 2025 Discovery of the Year award for its work on the BRAF protein and its role in the spread of cancer cells. The winner was chosen by the public, with more than 2,000 people participating in the vote.
![]()
-
March 31, 2026 In Search of New Antibiotics to Counter Antibiotic Resistance
IRIC’s Drug Discovery Unit (DDU) is part of the project “Designing Antibiotics Targeting Klebsiella Using Artificial Intelligence,” which has received US$3.8 million (approximately CA$5.3 million) from the Gates Foundation.
![]()
-
March 25, 2026 A model for modern biopharma collaboration — the enduring partnership between Ipsen, IRIC and IRICoR
In an era where breakthrough science increasingly depends on deep, cross sector collaboration, the longstanding partnership between Ipsen and the teams at the Institute for Research in Immunology and Cancer (IRIC) and its commercialization unit, IRICoR, at Université de Montréal stands out as a model for how academia and industry can accelerate science with purpose. First initiated in 2020 and now extended twice, the partnership is now collaborating on four active oncology research programs. Pierre Beaulieu, Associate Director, Drug Discovery Unit at IRIC, Hugo Lavoie, Associate Director of the Intracellular Signalling Research Unit at IRIC, and Chris Hupp, Senior Director, Research & External Innovation at Ipsen, explain how this collaboration has evolved from a promising research alliance into a formidable engine to deliver transformative therapies for patients.
![]()
-
March 23, 2026 Meet Simon-Pierre Gravel, new Principal Investigator at IRIC!
IRIC is proud to announce the arrival of Simon-Pierre Gravel as the new Principal Investigator in the Cancer metabolism and signaling Unit. Simon-Pierre Gravel has kindly agreed to answer a few questions
![]()
-
March 11, 2026 Patients and scientists: working together to fight blood cancers!
On Thursday, February 25, IRIC welcomed patients with leukemia or lymphoma for a unique opportunity for students and scientists at the Institute to share experiences: a chance to shine a spotlight on people with cancer at the heart of research.
![]()
-
February 26, 2026 Funding to make bioinformatics research at IRIC more environmentally responsible
IRIC’s information technology (IT) team has obtained funding from the Vice-Rectorate for Research and Innovation of the Université de Montréal to replace outdated cooling units with more energy-efficient models.
![]()
-
February 11, 2026 Trang Hoang, champion of human-centered scientific research
At IRIC, everyone knows researcher Trang Hoang. One of the Institute’s founders, she has headed its Hematopoiesis and Leukemia Research Unit since 2003. Her commitment, passion and talent for collaboration have benefited multiple generations of researchers and students over the years. On this International Day of Women and Girls in Science, let’s take a moment to shine a spotlight on the career of this trailblazer.
![]()
-
February 10, 2026 Félicitations aux récipiendaires du Concours de Bourses de maitrise et doctorat de l’IRIC!
Congratulations to the recipients of the IRIC Master’s and Doctoral Scholarship Contest!
![]()
-
February 4, 2026 UM171 Cell Therapy: Science Meets Humanity
On World Cancer Day, Anne Marinier, Director of IRIC’s Drug Discovery Unit, talks with Pasquale Gentile, a patient cured by the UM171 cell therapy developed at IRIC. A rich and deeply human encounter that gives hope to cancer research.
![]()
-
January 21, 2026 Delphine Bouilly recognized by the Royal Society of Canada and the Government of Quebec
Professor Delphine Bouilly, who heads IRIC’s Design and Application of Electronic Nanobiosensors Research Unit, has earned two prestigious awards in recent months for her outstanding research work. Delphine Bouilly was a finalist for the Hubert Reeves Award at the 2025 Prix du Québec. She was also appointed to the College of New Scholars, Artists and Scientists of the Royal Society of Canada (RSC). These honors add to an impressive list of achievements that has continued to grow since the researcher was recruited in 2017.
![]()
-
January 8, 2026 IRIC breakthrough among Québec Science’s top 10 discoveries of the year
The team led by Marc Therrien, Executive Director and Principal Investigator at IRIC, has solved a fundamental mystery in cancer biology: it has uncovered how a dysregulated protein allows cancer to proliferate in the human body. This scientific breakthrough, published in the journal Science, is one of the 10 most remarkable scientific discoveries of 2025 selected by the jury of Québec Science magazine.
![]()
-
December 15, 2025 Making breast cancer cells more visible to the immune system
The team led by Pierre Thibault, Director of IRIC’s Proteomics and Mass Spectrometry Research Unit, has discovered that abemaciclib, used to treat hormone receptor-positive metastatic breast cancer, reprograms the antigenic profile of tumors—a characteristic that could be exploited to improve treatment efficacy. Led by doctoral student Robin Minati, this study has just been published in the journal Cell Reports. It is the result of a close collaboration with the team of Claude Perreault, Director of IRIC’s Immunobiology Research Unit.
![]()
-
November 18, 2025 A novel role for pH in protein regulation
The team led by Sylvain Meloche, Director of IRIC’s Signalling and Cell Growth Research Unit, has discovered a new mechanism for the global regulation of proteins by intracellular pH. The study, co-led by PhD students Chloé Tesnière and Fadia Boudghene-Stambouli, has been published in the journal PNAS. The teams led by Pierre Thibault, Director of IRIC’s Proteomics and Mass Spectrometry Research Unit, Laurent Counillon, Director of the Molecular Physio-Medicine Laboratory at CNRS-Université Côte d’Azur (Nice, France), and Stine F. Pedersen d of the University of Copenhagen also contributed to the project.
![]()
-
November 14, 2025 National Philanthropy Day: Giving Youth a Voice!
November 15, 2025, is National Philanthropy Day across Canada. This day is an opportunity to honor philanthropists and their impact on society. IRIC has chosen to highlight the recent achievements of the IRIC Young Philanthropists Committee and some of the faces behind the committee’s success. IRIC is proud to introduce two of its founding members: Katrina Kontaxis and Benjamin Raynauld.
![]()
-
November 11, 2025 A colorful pickleball tournament for Young Philanthropists to benefit IRIC!
On October 26, IRIC’s Young Philanthropists held their eagerly awaited event at the Atwater Club. On the agenda? A pickleball tournament and a distinguished brunch! It was a bold and perfectly executed formula that brought together nearly 150 participants from Montreal’s next generation, all present for the cause.
![]()
-
November 5, 2025 An IRIC team attends the event “Light The Night”
On October 25, a team of nearly thirty people from IRIC took part in the “Light the Night” walk organized by the Leukemia & Lymphoma Society of Canada.
![]()
-
November 3, 2025 Congratulations to the winners of the 2025 Concours science réflexion !
IRIC is pleased to announce the winners of the concours Science Réflexion 2025. This annual contest highlights the creativity and talent of the IRIC community.
![]()
-
October 29, 2025 Delphine Bouilly and Madline Sauvage: improving breast cancer monitoring and treatment
Madline Sauvage, a doctoral student in molecular biology, and Professor Delphine Bouilly, Director of IRIC’s Design and Application of Electronic Nanobiosensors Research Unit, are recipients of a Pierre-Péladeau grant. Their start-up, SensÈn, whose mission is to make a clinical difference for breast cancer patients, won the second prize of $50,000 at the 27th edition of the competition. These grants, offered since 1999, aim to support the next generation in all fields of study in starting businesses, developing business plans, and developing products and markets.
![]()
-
October 29, 2025 Eralda Kina: Combining Research and Clinical Practice
Eralda Kina is an oncologist and doctoral student in the laboratory of Claude Perreault, Principal Investigator in IRIC’s Immunobiology Research Unit. Claude Perreault’s team studies the cells that regulate the functioning of the immune system, known as “T lymphocytes,” in order to understand and improve the functioning of the immune system. The team’s ultimate goal is to develop therapeutic vaccines against cancer.
![]()
-
October 29, 2025 Interview with Sylvie Mader - Studying breast cancer: interconnected motivations
The team led by Sylvie Mader, Principal Investigator in IRIC’s Molecular Targeting in Breast Cancer Research Unit, is working to understand the causes of breast cancer subtype diversity, identify new targets for drug development, and understand the causes of resistance to molecules used to treat breast cancer.
![]()
-
October 29, 2025 Interview with Geneviève Deblois - Focus on breast cancer
The team led by Geneviève Deblois, Principal Investigator in IRIC’s Metabolic and Epigenetic Alterations in Cancer Research Unit, is studying a particularly aggressive subtype of breast cancer called triple-negative breast cancer, which is characterized by high relapse rates and a lack of targeted therapeutic options. Patients with this type of cancer are usually treated with chemotherapy, but relapse rates are high. The team is seeking to understand how these tumor cells adapt and survive in a microenvironment depleted of nutrients and oxygen, as well as the mechanisms that enable them to resist chemotherapy, in order to develop new, more effective therapeutic strategies.
![]()
-
October 29, 2025 2025 IRIC Symposium : Spatial Biology from Every Angle
On October 24, IRIC held an international scientific symposium to explore the capabilities of spatial biology techniques. More than 150 members of the broader IRIC research community, from Montreal and beyond, gathered to discuss the latest advances in spatial sequencing, computational pathology, and analytical pipelines.
![]()
-
October 20, 2025 Running for a cause: to each their own pace!
In early summer 2025, Mira-Sue Mallet, a human resources partner at IRIC, joined the IRIC Running Club for the first time. What was supposed to be just an opportunity to get in shape and have fun with colleagues turned into a real accomplishment. Not only did Mira-Sue lend a hand to raise funds for research, she also committed to her first 5K at the Beneva Montreal Marathon, which she proudly completed! IRIC is proud to introduce an inspiring colleague who persevered to the end.
![]()
-
October 17, 2025 A major donation of $750,000 in support of cancer research
On October 1, four loyal IRIC donors, Danielle Lysaught, Paul Hamelin, Nigel Hamer and Jean-Marc Rousseau, received an IRIC lab coat, a symbolic gesture to thank them for their ongoing dedication to IRIC research.
![]()
-
October 16, 2025 Sébastien Lemieux and Carino Gurjao receive funding from the Canada Foundation for Innovation (CFI) and the Government of Quebec
Sébastien Lemieux, Principal Investigator and Scientific Director of IRIC’s
Bioinformatics Core Facility, together with Carino Gurjao, Principal Investigator at the Institute’s Genomic and Integrative Medicine Research Unit, are recipients of a research grant from the Canada Foundation for Innovation (CFI) through the John R. Evans Leaders Fund (JLEF) for their project “Infrastructure for the Development of Computational Approaches to Cancer Research,” for the period from March 2025 to April 2027.![]()
-
October 16, 2025 IRIC on the road again this upcoming fall 2025!
As part of its IRIC on the road student recruitment tour, the Institute’s academic affairs team will participate this fall in several graduate studies fairs organized by Canadian universities.
![]()
-
October 10, 2025 The Quebec Clinical Research Club awards an honorary prize to Michel Bouvier
At the annual meeting of the Quebec Clinical Research Club (CRCQ), Michel Bouvier was awarded the 2025 Scientific Mentor Award. This honor was bestowed upon him in recognition of his exceptional qualities as a mentor, his commitment to numerous students, the quality of his teaching, and his passion for nurturing the next generation.
![]()
-
October 8, 2025 Meet Edouard Williams Brunet: a young ambassador dedicated to the cause!
IRIC had the opportunity to speak with Edouard William Brunet, co-chair of the Jeunes Philanthropes Committee of IRIC. Diagnosed with cancer in his early twenties, Edouard wants to give back by raising funds for research. IRIC is proud to highlight this inspiring individual.
![]()
-
October 6, 2025 Nearly $16,000 raised during the Beneva Montreal Marathon
The Beneva Montreal Marathon took place on the weekend of September 20. As a loyal partner of the event, IRIC raised nearly $16,000 for cancer research thanks to 150 generous contributors.
![]()
-
October 2, 2025 From idea to clinical trial: a scientific ultra-marathon
A major milestone was recently reached with the start of a phase 1 clinical trial for a small molecule therapy for solid tumors licensed to Ipsen. The molecule in question inhibits the RAF protein, which is part of the pro-tumor Ras-MAPK signaling pathway. This breakthrough was made possible by the synergy between the teams of Anne Marinier, Director of IRIC’s Drug Discovery Unit, and Marc Therrien, Director of the Intracellular Signaling Research Unit and Chief Executive Officer of IRIC.
A look back and explanation of the long-term work, initiated in fruit flies and spanning more than twenty years, which has led to the clinical phase:
![]()
-
September 25, 2025 Carino Gurjao secures funding to advance research on metastatic colorectal cancer
As part of a joint call for projects by Pfizer and Colorectal Cancer Canada, Carino Gurjao, Principal Investigator at IRIC’s Genomic and Integrative Medicine Research Unit, has been awarded nearly $100,000 for his project “A Nanopore Sequencing Approach for Real-time Precision Medicine.”
![]()
-
September 11, 2025 A major federal grant for Delphine Bouilly, Sylvie Mader and Sébastien Lemieux’s laboratories
Delphine Bouilly’s team, in collaboration with Sylvie Mader and Sébastien Lemieux, has been awarded major funding from the Government of Canada’s New Frontiers in Research Fund – Exploration Stream, for her project “Exploiting Electrolytic Gating in Graphene Field-Effect Nanosensors to Achieve Multiplexed Breast Cancer Profiling for Personalized Medicine”.
![]()
-
September 8, 2025 Two IRIC researchers receive funding from the Natural Sciences and Engineering Research Council of Canada (NSERC)
In 2025, as part of the Discovery Grants Program, NSERC awarded significant five-year funding to the team led by Philippe Roux, head of the Cell Signalling and Proteomics Research Unit, and to the team led by Matthew Smith, head of the Cancer Signalling and Structural Biology Research Unit.
![]()
-
September 2, 2025 IRIC and the Faculty of Pharmacy at the Université de Montréal recruit Olivier Mailhot as Principal Investigator
IRIC is proud to announce the arrival of Olivier Mailhot as a new Principal Investigator in the Computational Drug Discovery Research Unit. He will hold the position of assistant professor in the Faculty of Pharmacy at the Université de Montréal (UdeM). Olivier Mailhot has also been appointed IVADO professor.
![]()
-
September 2, 2025 Meet Olivier Mailhot, new Investigator at IRIC
IRIC is proud to announce the arrival of Olivier Mailhot as a new Principal Investigator in the Computational Drug Discovery Research Unit. Professor Mailhot generously agreed to answer a few questions:
![]()
-
August 28, 2025 Marc Therrien elected member of the Canadian Academy of Health Sciences
Marc Therrien, Principal Investigator in the Intracellular Signalling Research Unit and Chief Executive Officer of IRIC, as well as Full Professor in the Department of Pathology and Cell Biology at the Faculty of Medicine of the Université de Montréal, joins the Canadian Academy of Health Sciences (CAHS) as a new member. A total of 47 new members have been elected to the CAHS for the year 2025.
![]()
-
August 22, 2025 IRIC highlights the scientific knowledge of the next generation!
The 7th edition of IRIC’s 2025 Summer Interns’ Scientific Day was held on Thursday, August 7.
![]()
-
August 12, 2025 Behind-the-scenes work brought to light: CIHR award for investigators Gregory Emery and David Knapp
Gregory Emery and David Knapp, directors of the IRIC Vesicular Trafficking and Cell Signalling Research Unit and Cellular Engineering Research Unit, respectively, have both received a special mention for outstanding contribution from the Canadian Institutes of Health Research (CIHR) College of Reviewers. In addition, Gregory Emery was recognized for his outstanding evaluation services during three or more consecutive project grant competitions, a distinction awarded to only 1% of evaluators nationwide. David Knapp received three citations for outstanding contribution. This recognition reflects the remarkable quality of their evaluation work and their constructive participation in committee discussions.
![]()
-
July 29, 2025 Unmutated tumor antigens: exploitable targets for immunotherapy
The team headed by Claude Perreault, Director of IRIC’s Immunobiology Research Unit, has identified novel tumor antigens that could lead to the development of vaccines for the treatment of two cancers: melanoma and non-small cell lung cancer. To achieve this, the team led by PhD student Anca Apavaloaei has adopted an unbiased approach that examines all genetic material, including DNA described as “junk” because it is not coding for any known protein. The work, the fruit of collaborations with the Epitopea company, McGill University, the University of Liège and the University of Lausanne, has been published in the journal Nature Cancer.
![]()
-
July 22, 2025 Julie Lessard and David Knapp awarded in CIHR’s latest competition
The Canadian Institutes of Health Research (CIHR) recently awarded Project Grants for the Spring 2025 competition. This program aims to support original ideas with the potential to advance basic and applied health knowledge. One project involving IRIC teams is a recipient of these prestigious grants.
![]()
-
July 22, 2025 Running: a mirror of life. Testimony of Sasmita Tripathy, Assistant Director of IRIC’s Drug Discovery Unit
Sasmita Tripathy has been a runner for many years, a sport she loves for both her physical and psychological well-being. Through her testimony, she shows us how all spheres of her life are interrelated; a touching reflection that highlights her determination, her kindness and her will to change things, both in the laboratory and in her personal life. IRIC is proud to present this inspiring personality.
![]()
-
July 10, 2025 New genetic tools to study chromatin remodeling in hematopoiesis
The team led by Julie Lessard, Director of IRIC’s Chromatin Structure and Stem Cell Biology Research Unit, has generated new genetic tools to study the role of two subunits of the SWI/SNF chromatin remodeling complexes in hematopoiesis. The work, led by research advisor Pierre Priam, is published in the journal Experimental Hematology.
![]()
-
July 9, 2025 2025 Student Recruitment Event: Meet the next generation of inspiring scientists!
Each year, the Student Recruitment Event offers to students interested in pursuing graduate studies in cancer research a unique opportunity to participate in an international recruitment event at the Institute for Research in Immunology and Cancer of the Université de Montréal. The 2025 edition took place from June 3 to 6 and welcomed 31 participants from various universities around the world. For this edition, more than 139 interviews were scheduled between participants and researchers from the Institute attending the event. During their stay, participants were able to:
![]()
-
July 7, 2025 Stem Cell Network funds two projects by Guy Sauvageau’s team
The Stem Cell Network (SCN) recently awarded $13.5 million to fund 36 regenerative medicine research projects and clinical trials across the country. Guy Sauvageau, Director of the Molecular Genetics of Stem Cells Research Unit, has received funding for two projects through these grants.
![]()
-
July 4, 2025 Six Fonds de recherche du Québec scholarships and grants awarded to IRIC members for 2025-2026.
The Fonds de recherche du Québec (FRQ) is offering $38.2 million in training awards, $13.6 million in career awards and $10.8 million in research grants for a total of $62.6 million in new awards for 2025-2026.
![]()
-
July 2, 2025 6.5 million in funding to understand and target ovarian cancer cell dormancy
The Canadian Cancer Society (CCS) recently announced the results of the 2025 Breakthrough Team Grants: Transforming the Future of Metastatic Cancer competition. Philippe Roux, Scientific Director and Director of IRIC’s Cell Signalling and Proteomics Research Unit, is part of a multidisciplinary, multi-centre team receiving $6.5 million in funding to understand and target ovarian cancer dormancy.
![]()
-
June 25, 2025 Student researchers honoured at IRIC’s 11th Scientific Day!
The 11th Scientific Day, held on June 12, highlighted the work of IRIC’s scientific community. Nearly 190 people attended and had the opportunity to attend nine oral presentations, six flash presentations, and 50 poster presentations. To highlight the knowledge of the next generation of scientists, seven poster presentation awards and two oral presentation awards were presented.
![]()
-
June 13, 2025 Success for IRIC at the Festival Eurêka! 2025
On May 30, live from Île Sainte-Hélène, IRIC took part in the 18th edition of the Festival Eurêka! This festival is an opportunity for families and up-and-coming young people to immerse themselves in the world of scientific discovery.
![]()
-
June 10, 2025 A Coup de Coeur for Éric Bonneil at the 2025 Rector’s Awards at the Université de Montréal
The Université de Montréal’s Rector’s Awards 2025 were presented on May 29 to highlight the remarkable contributions of staff members whose commitment and achievements exemplify the values of the University. IRIC is proud to recognize Éric Bonneil, Manager of the Proteomics Core Facility, as Coup de Coeur in the Innovation category.
![]()
-
June 4, 2025 Meet Doris Alexandra Schuetz, Principal Research Advisor, Drug Discovery Unit
Doris Alexandra Schuetz is an inspiration to many members of the IRIC community. A committed professional dedicated to the fight against cancer on a daily basis, Doris also excels at running, a sport she has been practising for several years now. Eager to combine business with pleasure, Doris has raised close to $5,000 for cancer research at IRIC since 2022, through her participation in the Montreal Beneva Marathon. IRIC is proud to introduce you to this inspiring person.
![]()
-
May 29, 2025 BRAF mutants escape stopped by therapeutic “agents”
The team led by Marc Therrien, CEO and Director of IRIC’s Intracellular Signaling Research Unit, has identified major structural changes that enable the BRAF protein to escape control and mimic its activation, giving it strong oncogenic potential. What’s more, the researchers succeeded in completely restoring the protein’s normal conformational state using small molecules – a breakthrough. The study, published in the prestigious journal Science, is the joint work of Hugo Lavoie (Associate Director), Ting Jin (Research Advisor) and Driss Lajoie (Research Advisor), who are the co-first authors. It is part of a close collaboration between several research teams which, in addition to the Therrien laboratory, involves teams from NYU Langone (New York), Université de Bordeaux and the University of Calgary.
![]()
-
May 23, 2025 Next Generation Competition Program scholarship recipients arrive at the Institute
This summer, 10 students have the opportunity to complete a 12- or 16-week research internship at IRIC, in addition to benefiting from a scholarship of excellence.
![]()
-
May 21, 2025 Jean-Coutu and Marcelle-Coutu pavilions: 20 years ago, a dream came true
On April 26, 2005, the two pavilions were inaugurated to house the Université de Montréal’s Faculty of Pharmacy and Institute for Research in Immunology and Cancer (IRIC), respectively.
![]()
-
April 30, 2025 A total envelope of $612,610 for a project led by Sylvain Meloche, with support from IRIC, IRICoR, Immune Biosolutions and the SRC
In early 2025, Quebec’s Ministère de l’Économie, de l’Innovation et de l’Énergie (MEIE) awarded $475,000 in funding to Sylvain Meloche’s team, which heads IRIC’s Signalling and Cell Growth Research Unit. Axelys supported the team in the preparation of this funding application under the Programme de Maturation technologique (PSOv2d). This funding leverages additional private funding from Immune Biosolutions and the Société de Recherche sur le Cancer (SRC) respectively.
![]()
-
April 15, 2025 IRIC Gala Audace: $1,060,025 raised for cancer research
On April 9, the 2025 edition of the Gala Audace of the Institute for Research in Immunology and Cancer (IRIC) of the Université de Montréal was held. Thanks to the generosity of over 300 guests at the Marché Bonsecours, our valued donors and sponsors, a total of $1,060,025 was raised to support cancer research at IRIC.
![]()
-
March 26, 2025 Manipulating the very small at large scale
The team led by Delphine Bouilly, director of IRIC’s Electronic Nanobiosensor Design and Application Research Unit, has developed an original technique for manipulating and moving graphene, a one-atom-thick material involved in the assembly of electronic chips, for applications in molecular diagnostics. Led by doctoral student Amira Bencherif, the study was published in the journal npj 2D materials and applications.
![]()
-
March 3, 2025 Meet Carino Gurjao, new Principal Investigator at IRIC
IRIC is proud to announce the arrival of Carino Gurjao as the new Principal Investigator of the Genomic and Integrative Medicine Research Unit.
Professor Gurjao generously agreed to answer a few questions:
![]()
-
March 3, 2025 IRIC and UdeM recruit Carino Gurjao as Principal Investigator
IRIC is proud to announce the arrival of Carino Gurjao as a new Principal Investigator in the Genomic and Integrative Medicine Research Unit. He will hold the position of Assistant Professor in the Department of Medicine at Université de Montréal (UdeM), affiliated with IVADO.
![]()
-
February 19, 2025 Celebrating 20 years of transplantation for William Brock!
February 17, 2025, will remain a milestone for William Brock, Chairman of IRIC’s Board of Directors. It’s been 20 years since he received a stem cell transplant that cured him of leukemia… a transplant that changed his life forever!
![]()
-
February 14, 2025 Canadian Institutes of Health Research funds five research projects at IRIC
The Canadian Institutes of Health Research (CIHR) recently awarded Project Grants for the Fall 2024 competition. This program aims to support original ideas with the potential to advance basic and applied health knowledge.
Across the country, 453 research projects were funded, representing a total investment of $411 million. Five projects involving IRIC teams are recipients of these prestigious grants.
![]()
-
February 11, 2025 A student committed to enhancing the representation of women and girls in science!
On this International Day of Women and Girls of Science, IRIC had the opportunity to talk to Fanny-Meï Cloarec-Ung, a doctoral student in David Knapp’s laboratory. Committed to enhancing the representation of women in the sciences, Fanny-Meï stands out for her leadership and her willingness to make a real difference.
![]()
-
January 28, 2025 Ludovic Tamaro: the story of a young patient saved by research
On this World Cancer Day, meet Ludovic Tamaro, a 24-year-old diagnosed with acute myeloid leukemia and myelodysplasia when he came of age.
![]()
-
January 23, 2025 A picture is worth a thousand words - or why cell painting technology holds promise for drug discovery
IRIC’s high-throughput screening platform has been awarded $583,654 by Quebec’s Ministère de l’Économie, de l’Innovation et de l’Énergie, following a call for projects launched as part of the Quebec Research and Innovation Investment Strategy 2022-2027 (SQRI2). The funded project aims to integrate real-time cell painting and artificial intelligence-based phenotypic classification technology for the discovery of new drugs.
![]()
-
January 22, 2025 Sébastien Lemieux takes over as Scientific Director of Calcul Québec
Sébastien Lemieux, Director of IRIC’s Functional and Structural Bioinformatics Research Unit, was appointed Scientific Director of Calcul Québec last November.
![]()
-
January 15, 2025 Identification of promising small molecules for the treatment of pediatric cancers
The team led by Brian Wilhelm, Director of IRIC’s High-Throughput Genomics Research Unit, has developed a new drug discovery platform for high-risk leukemias. Doctoral student Safia Safa-Tahar-Henni led the project, which has been published in the journal Leukemia.
![]()
-
January 9, 2025 Millénium Québécor’s Recherche Ton Impact program rewards doctoral student Madline Sauvage for her start-up company.
Madline Sauvage, a doctoral student in the laboratories of Delphine Bouilly and Sylvie Mader at IRIC, has been awarded a $6,000 bursary from the Recherche ton Impact entrepreneurial training of the Université de Montréal’s Millénium Québécor program.
![]()
-
January 7, 2025 Major funding for the creation of immunotherapies for the laboratories of Claude Perreault and Pierre Thibault
Thanks to a major grant of nearly $1.5 million from the Quebec government, CQDM and Epitopea are able to fund a high-potential collaborative research project at the heart of the Université de Montréal, directly involving the research teams of Claude Perreault and Pierre Thibault, principal investigators at IRIC. The aim of the project is to develop a new immunotherapy to effectively treat patients with lung and ovarian cancers.
![]()
-
December 16, 2024 Dr Vincent Quoc-Huy Trinh’s laboratory receives major funding
The John R. Evans Leaders Fund, the Canada Foundation for Innovation and the research support program of Quebec’s ministère de l’Économie, de l’Innovation et de l’Énergie have awarded Dr. Vincent Quoc-Huy Trinh’s lab significant funding for its promising research project: “The effect of fibroblasts on the malignant transformation of dysplastic stem cells in pancreatic tumors”, for the period 2024-2026.
![]()
-
December 9, 2024 Multinational consortium led by Julie Lessard awarded major grant
As part of TRANSCAN-3’s joint transnational call 2023 for translational research on cancer epigenetics, Julie Lessard has been awarded a $2,03M grant for the multinational consortium she leads, alongside 5 colleagues from France, Spain and Israel.
![]()
-
November 20, 2024 Two physiological responses to protect tissues from nuclear defects
A new study by the Cell Cycle Regulation Research Unit, headed by Vincent Archambault, identifies two physiological responses activated when nuclear reformation defects are detected by the cells at the end of their division. These responses play a crucial role in protecting tissue integrity during development. Published in the journal PLOS Biology, the project was led by doctoral student Jingjing Li.
![]()
-
November 18, 2024 Together for the Cause!
On September 21 and 22, the Beneva Marathon took place in Montreal, and IRIC is proud to announce that nearly $33,500 was raised, with a total of 454 donations. This significant contribution placed IRIC in 5th position among all participating organizations.
![]()
-
November 12, 2024 Major Research Grants at IRIC
It is with great pleasure that IRIC highlights the recipients of major grants offered by partner organizations. A well-deserved recognition for the Institute’s research teams, who certainly have the wind in their sails!
![]()
-
November 6, 2024 Announcement of the winners of the Science Réflexion 2024 contest
IRIC is pleased to announce the winners of the Science Réflexion 2024 contest. This annual contest highlights the creativity and talent of the IRIC community and presents science in a new way.
![]()
-
November 6, 2024 Challenge: to create an advanced materials academy
Delphine Bouilly, Université de Montréal’s representative on the executive of the Regroupement québécois sur les matériaux de pointe (RQMP), and her colleagues will set up the Advanced Materials Academy (AMA), a new initiative to enrich graduate training in advanced materials. The project is made possible by a $1.65M grant from the Natural Sciences and Engineering Research Council of Canada’s (NSERC) CREATE (Collaborative Research and Training Experience) program.
![]()
-
October 21, 2024 A CRS Doctoral Research Awards winner at IRIC!
IRIC would like to congratulate Anaïs Vivet on receiving the Doctoral Research Awards from the Cancer Research Society (CRS). She is one of 40 recipients of this second edition of the Doctoral Research Awards.
![]()
-
October 16, 2024 Improving the design of experiments involving BRET biosensors to make them more physiologically relevant
A new study by the laboratory of Michel Bouvier, Director of IRIC’s Molecular Pharmacology Research Unit, compares the advantages and disadvantages of various biosensors used to analyze G protein-coupled receptor signaling. The research project was conducted jointly by postdoctoral fellow Shane Wright, research advisor Charlotte Avet and postdoctoral fellow Supriya Gaitonde. It is published in the journal Science Signaling.
![]()
-
October 3, 2024 Anca Apavaloaei receives the 2024 Best Thesis Award
![]()
-
October 2, 2024 Two students determined to walk for leukemia
The annual Light The Night walks, organized by the Leukemia & Lymphoma Society of Canada, brings together the blood cancer community across the country. The event raises funds for research on 137 types of blood cancers. Students Ryan Borotra and Tyler Lussier have mobilized a team for IRIC, and will be supporting the cause during the Montreal Light The Night walk on October 26. Meet these two committed representatives of the next generation.
![]()
-
September 18, 2024 Development of a tool to study new treatments for liver cancer
The team led by Sylvain Meloche, Director of IRIC’s Signalling and Cell Growth Research Unit, has developed a new preclinical model for the study of the most common subtype of liver cancer. Published in the journal Disease Models & Mechanisms, the work was carried out by research associate Laure Voisin and research advisor Marjorie Lapouge. Vincent Quoc-HuyTrinh, Director of IRIC’s Digital Histology and Advanced Pathology Research Unit, also contributed to the study.
![]()
-
September 10, 2024 Katherine Borden elected Fellow of the Canadian Academy of Health Sciences
Katherine Borden, Principal Investigator and Director of the Structure and Function of the Cell Nucleus Research Unit at IRIC, and Full Professor in the Department of Pathology and Cell Biology in the Faculty of Medicine at Université de Montréal, is one of XX new Fellows elected to the Canadian Academy of Health Sciences (CAHS).
![]()
-
September 9, 2024 IRIC on the road again this upcoming fall
As part of its IRIC on the road student recruitment tour, the Institute’s academic affairs team will participate this fall in several graduate studies fairs organized by Canadian universities.
![]()
-
September 4, 2024 Genetic parasites involved in the development and function of the immune system?
A recent study by the team of Claude Perreault, Director of IRIC’s Immunobiology Research Unit, proposes three potential functions for “parasite” DNA sequences in the development of T lymphocytes. Led by doctoral student Jean-David Larouche, the work is published in the journal eLife.
![]()
-
July 31, 2024 A novel approach to study hypoxia enables identification of a potential new marker for ovarian cancers
In a new study, the team led by Etienne Gagnon, Director of IRIC’s Cancer Immunobiology Research Unit, has developed a cell culture protocol that accurately reproduces the characteristic conditions of primary tumors. The group also identified a novel form of the WT1 protein associated with poor long-term survival in ovarian cancer patients. Published in the journal Cancer Gene Therapy, the study was led by doctoral student Jordan Quenneville.
![]()
-
July 30, 2024 All but nanometric leadership for Delphine Bouilly
Delphine Bouilly, Director of IRIC’s Design and Application of Electronic Nanobiosensors Research Unit, has been on a roll in recent months. Appointed head of three strategic networks, she has also seen her Canada Research Chair in Bionoelectronics renewed and won a prestigious award from the Electrochemical Society. Here’s a look back at her record of achievement.
![]()
-
July 29, 2024 Three IRIC graduates named to the Honor List of the Rector of the Université de Montréal
IRIC is proud to count three of its graduates among the most recent appointments to the Rector’s Honor List.
![]()
-
July 25, 2024 Sébastien Carréno collaborates with a team from France on a study published in EMBO J
Sébastien Carréno, Director of IRIC’s Cellular Mechanisms of Morphogenesis during Mitosis and Cell Motility Research Unit, has signed a new publication in EMBO J with colleagues from the Institute of Pharmacology and Structural Biology of Toulouse. The project was led by PhD student Perrine Verdys, co-supervised by Professor Carréno. The group’s work demonstrates that macrophages, which are immune cells, can migrate without the ERM proteins, cell cortex proteins until now considered essential to any cell migration process.
![]()
-
July 23, 2024 Three IRIC projects funded by the Canadian Institutes of Health Research
The Canadian Institutes of Health Research (CIHR) recently awarded Project Grants for the Spring 2024 competition. This program aims to support original ideas with the potential to advance basic and applied health knowledge. Across the country, 373 research projects, representing a total investment of $325 million, have been funded. Three IRIC projects are recipients of these prestigious grants.
![]()
-
July 4, 2024 2024 Student Recruitment Event: meeting a new generation of motivated and diversified students
The 2024 edition took place from June 5 to 7 and welcomed 32 participants from universities around the world. More than 150 interviews were scheduled this year between participants and the Institute researchers present at the event.
![]()
-
June 25, 2024 Targeting the surface of leukemia cells with immunotherapies
Immunotherapies, which are more natural and less toxic than chemotherapies, have made major advances in recent years. However, they remain relatively unexploited in the treatment of acute myeloid leukemia (AML), a type of leukemia that is particularly heterogeneous genetically and difficult to treat. The Leucegene group, led by Guy Sauvageau (IRIC’s Molecular Genetics of Stem Cells Research Unit) and Josée Hébert (Centre de recherche de l’Hôpital Maisonneuve-Rosemont), and involving the teams of Philippe Roux, Sébastien Lemieux and Pierre Thibault at IRIC, and of Vincent-Philippe Lavallée at the CHU Ste-Justine Research Centre, has developed a new approach to analyze the molecules present on the surface of leukemia cells. Their work has led to the identification, in AML specimens, of new high-potential targets for immunotherapies. The study was led by Marie-Ève Bordeleau, Deputy Director of the Sauvageau laboratory, and is published in the journal Cell Reports.
![]()
-
June 13, 2024 Marc Therrien takes the position of Interim Chief Executive Officer of IRIC
IRIC is pleased to announce the appointment of Professor Marc Therrien as Interim Chief Executive Officer of IRIC. The Executive Council of the Université de Montréal recently ratified the recommendation made by IRIC’s Board of Directors. Marc Therrien is Principal Investigator in the Institute’s Intracellular Signalling Research Unit and Full Professor in the Department of Pathology and Cell Biology at the Faculty of Medicine of the Université de Montréal.
![]()
-
June 6, 2024 Cole Foundation scholarships for two IRIC students
Two IRIC students win Cole Foundation doctoral scholarships worth $50,000 for two years.
![]()
-
June 5, 2024 Welcome to the 2024 IRIC Next Generation Awards recipients!
This scholarship enables undergraduate students to spend 12 to 16 weeks at IRIC during the summer term.
![]()
-
June 5, 2024 IRIC Master’s and Ph.D. Scholarships – Recipients of the Winter 2024 Competition
The objective of the IRIC Master and Doctoral Awards Competition is to support IRIC students who have an excellent academic and research records, but do not benefit from substantial scholarship.
![]()
-
June 5, 2024 Success for IRIC at the Festival Eurêka! 2024
On May 24, live from Île Sainte-Hélène, IRIC took part in the 17th edition of the Festival Eurêka! This festival is an opportunity for families and the next generation of young people to immerse themselves in the world of scientific discovery.
![]()
-
June 4, 2024 From lab to stage, Vanessa Laflamme piles up the miles and dollars in support of research
A biologist by training, having completed a Master’s degree in Molecular Biology at IRIC, Vanessa worked for a few years in the biotech industry before returning to IRIC in 2020. Since then, she has been working as a research advisor in the Drug Discovery Unit headed by Anne Marinier. A member of Équipe IRIC for a second year, she generously agreed to share her running and fundraising experiences with us.
![]()
-
May 30, 2024 End of Michel Bouvier’s mandate as Chief Executive Officer of IRIC
IRIC marks the end of Michel Bouvier’s mandate as the Institute’s Executive Director since 2014.
![]()
-
May 13, 2024 Seventeen scholarships and grants from the Fonds de recherche du Québec awarded to IRIC members for 2023-2024
The Fonds de recherche du Québec (FRQ) offers $53.2 million in training grants, $13.7 million in career grants and $52.8 million in research grants for a total of $119.7 million in new grants for the year 2024-2025. IRIC is proud to count several of its members among the winners of these competitions.
![]()
-
May 10, 2024 Les Grandes Retrouvailles at IRIC: discover the world of fundamental research
As part of Les Grandes Retrouvailles of Université de Montréal, IRIC opened its doors to the general public on May 9, giving them an opportunity to discover the world of fundamental research. Over twenty people were on hand to learn about cancer research and discover the Institute.
![]()
-
May 9, 2024 $3.7M in funding for a research project on cellular rejuvenation
A collaborative research project between the teams of Guy Sauvageau, director of the IRIC Molecular Genetics of Stem Cells Research Unit, and Anne Marinier, CEO of biotech company RejuvenRx, recently received $3,697,199 in funding. The grant was made possible by investments from the Quebec Consortium for Drug Discovery (CQDM), the Quebec government, RejuvenRx, the Canadian Institutes of Health Research and the Université de Montréal Partenariat fund.
![]()
-
May 6, 2024 $21M in funding to tackle antimicrobial drug resistance
The Canadian Biomedical Research Fund (CBRF) and the Biosciences Research Infrastructure Fund (BRIF) are investing $21 million in a research project aimed at accelerating the discovery of new antibiotics to counter drug-resistant bacteria. Led by Yves Brun, professor in the Department of Microbiology, Infectious Diseases and Immunology and researcher at the UdeM Faculty of Medicine’s Centre for Biomedical Innovation, the project will involve various IRIC teams.
![]()
-
April 11, 2024 $750 000 raised during the IRIC Audace Gala at the Université de Montréal
On April 10, over 250 guests gathered for IRIC’s Gala Audace 2024. Thanks to the generosity of donors and guests, $750 000 was raised. Since 2014, more than $8 million has been raised through the Gala Audace, helping to perpetuate IRIC’s fundamental research and perfect cancer therapies.
![]()
-
April 2, 2024 Success for the scientific outreach evening Cancer and immunology
On March 28, four IRIC students participed in the game of science outreach at the BistroBrain: Cancer and Immunology event.
![]()
-
March 25, 2024 A new strategy based on copper overdose to treat AML
Despite the therapeutic advances of recent years in the treatment of acute myeloid leukemia (AML), this pathology remains associated with a poor prognosis. In a new study, the Leucegene group led by Guy Sauvageau, Director of IRIC’s Molecular Genetics of Stem Cells Research Unit, Anne Marinier, Director of IRIC’s Drug Discovery Unit, and Josée Hébert (Banque de cellules leucémiques du Québec (BCLQ), Maisonneuve-Rosemont Hospital Research Center) proposes a therapeutic strategy based on copper overdose as a treatment for a genetic subgroup of AML particularly sensitive to this approach. Led by Céline Moison and Deanne Gracias (biology), and Julie Schmitt and Réjean Ruel (chemistry), in collaboration with the BCLQ genetics laboratory team, this project has been published in the prestigious journal Science Advances.
![]()
-
March 21, 2024 CSMB honours Sylvie Mader and Guy Sauvageau
IRIC congratulates Sylvie Mader and Guy Sauvageau, recipients of awards from the Canadian Society for Molecular Biosciences (CSMB). Sylvie Mader, Director of IRIC’s Molecular Targeting in Breast Cancer Treatment Research Unit, is the 2024 recipient of the Jeanne Manery-Fisher Memorial award. Guy Sauvageau, Director of IRIC’s Molecular Genetics of Stem Cells Research Unit, is the recipient of the Arthur Wynne Gold Medal award.
![]()
-
March 5, 2024 Identification of an enzymatic activity required for the completion of cell division
Cell division, a process required for the survival of all species, enables the genetic material of a mother cell to be separated and shared between its two daughter cells. As many aspects of cell division remain poorly understood, the team led by Jean-Claude Labbé, Director of the Cell Division and Differentiation Research Unit, focused on one of its final stages, cytokinesis. The team identified a new protein, OSGN-1, as being required for the correct completion of cell division. The study, published in the journal PNAS, was led by research associate Eugénie Goupil and doctoral student Léa Lacroix.
![]()
-
February 20, 2024 IRIC marks International Day of Women and Girls in Science
On February 10, IRIC was present at the Centre des sciences de Montréal for Women and Girls in Science Day, to mark the International Day of Women and Girls in Science. This 7th edition of the event drew a record crowd, with over 3,300 curious onlookers on hand to discover different scientific disciplines.
![]()
-
February 5, 2024 Pasquale Gentile: “Without science, I wouldn’t have survived my cancer.”
February 4 was World Cancer Day. Watch the video of Pasquale Gentile, a patient who received UM171 cell therapy, a molecule discovered by Guy Sauvageau and Anne Marinier’s teams at IRIC.
![]()
-
February 1, 2024 The Canadian Cancer Society awards a grant to Geneviève Deblois
Geneviève Deblois, Director of IRIC’s Metabolic and Epigenetic Alterations in Cancer Research Unit, is the recipient of a Emerging Scholar Research Grant from the Canadian Cancer Society (CCS). Sixteen of these grants were awarded across the country in the 2023 competition to advance cancer research programs. Geneviève Deblois, the only researcher to be funded in Quebec, has been awarded $550,000 for her project “Finding better ways to treat triple-negative breast cancer”.
![]()
-
January 29, 2024 Expansion of hematopoietic stem cells: the mechanism of action of the UM171 molecule involves MYC regulation
The ex vivo expansion of hematopoietic stem cells (HSCs), an important advance for cell-based therapies, can lead to replicative and metabolic stresses; much as aging does. In their efforts to identify the best strategies for HSC expansion, the team led by Guy Sauvageau, Director of IRIC’s Molecular Genetics of Stem Cells Research Unit, has been investigating the molecular mechanisms induced by the UM171 molecule, which was jointly discovered in 2014 by his laboratory and the team led by Anne Marinier, Director of IRIC’s Drug Discovery Unit, and enables the ex vivo expansion of HSC derived from umbilical cord blood. Their most recent study, led by researcher Jalila Chagraoui and published in the journal Blood, reports that the UM171 molecule preserves HSCs from cell culture-induced stress by modulating the activity of the MYC protein.
![]()
-
January 25, 2024 Decoding the molecular and structural bases of signal transduction to design more effective drugs
G protein-coupled receptors (GPCRs) are bound by a wide range of ligands: over 500 molecules naturally present in humans (such as hormones), as well as a third of approved drugs, target them. The binding of these ligands to GPCRs leads to the selective activation of different signaling pathways, which are involved in processes as varied as metabolism, cell growth and immune responses. Michel Bouvier, Director of the Molecular Pharmacology Research Unit and Chief Executive Officer of IRIC, and his collaborators set out to better understand, at the atomic level, the molecular and structural determinants that dictate a ligand’s ability to activate a GPCR. The work, a collaboration with Madan Babu, researcher at Ste-Jude Research Hospital, and Brian Kobilka, researcher at Stanford University and co-recipient of the 2012 Nobel Prize in Chemistry, was conducted by Franziska Heidenreich while she was a postdoctoral fellow in the Bouvier laboratory. It is published in the journal Science.
![]()
-
January 23, 2024 A molecular glue that promotes cyclin K degradation
The CDK12 and cyclin K proteins form a complex that plays important roles in several types of cancer, including melanoma and breast cancer. The team led by Philippe Roux, IRIC’s Scientific Director and Head of its Cell Signaling and Proteomics Research Unit, has characterized the mode of action of a novel CDK12 inhibitor, developed for its therapeutic anticancer potential. The results obtained reveal that the inhibitor in question, the molecule SR-4835, acts as a molecular glue to induce the degradation of cyclin K and thus inactivate the complex it forms with CDK12. Conducted jointly by postdocs Thibault Houles and Jonathan Boucher, the project has been published in the journal Cell Death Discovery.
![]()
-
January 15, 2024 A look back at IRIC’s 20th anniversary: A year of celebration!
Over the past 20 years, IRIC has built a research model unique in the country, and brought together exceptional expertise. To shine the spotlight on its community, promote science and set the stage for future achievements, the Institute has created a special program for 2023. Here, in retrospect, are the various initiatives and celebrations that marked this special year.
![]()
-
January 10, 2024 New study identifies targets for immunotherapy in two breast cancer subtypes
Breast cancer is the most common cancer in the world. There are several subtypes that differ immunologically, some generating stronger immune responses than others. The team led by Claude Perreault, Director of IRIC’s Immunobiology Research Unit, used a proteogenomic approach to study the immunopeptidome of two breast cancer subtypes: hormone receptor-positive breast cancer and triple-negative breast cancer. They identified several new antigens for each subtype, which were also shown to generate an immune response in vitro. These new antigens therefore represent potential targets for cancer immunotherapy. The project was led by doctoral student Eralda Kina and is published in The Journal of Clinical Investigation.
![]()
-
December 20, 2023 A $6M grant to improve the treatment of acute myeloblastic leukemia
Guy Sauvageau, as director of the IRIC Molecular Genetics of Stem Cells Research Unit, and Anne Marinier, as CEO of the biotechnology company RejuvenRx, obtain $6M over 3 years from Genome Canada for the project “Development of a Novel Cyclin K Degrader of High-risk AML Patients and Associated Genomic Features”. The funding is provided as part of Genome Canada’s Genomics Applications Partnership Program, which aims to foster partnerships between industry, public users, and academic research.
![]()
-
December 15, 2023 Two CRS Doctoral Research Award winners at IRIC!
IRIC would like to congratulate Myriam Iliana Ibanez Rios and Karla Lucia Paez Martinez on receiving a Doctoral Research Award from the Cancer Research Society (CRS). They are among 21 recipients of these new fellowships across the country, which offer funding of $70,000 over two years. This new CRS initiative aims to provide an essential support to the doctoral community.
![]()
-
December 14, 2023 Two UdeM Excellence Scholarship recipients among our student community!
The Assistant Vice-Rector for Graduate and Postdoctoral Studies honored student excellence by awarding 155 scholarships. Two IRIC members are among the recipients of these scholarships for excellence. Scholarships are awarded on the basis of academic merit, perseverance and volunteer work.
![]()
-
December 1, 2023 The Pillars of IRIC: meet Mélanie Fréchette
In 2023, IRIC celebrates its 20th anniversary. Among the initiatives put in place to celebrate this anniversary, the series of portraits entitled “The Pillars of IRIC” will highlight the people who have contributed since the very beginning, sometimes in the shadows, to make IRIC what it has become.
Today, meet Mélanie Fréchette, Animal Health Technician in Guy Sauvageau’s laboratory.
![]()
-
December 1, 2023 The Pillars of IRIC: meet Sandra Weber
In 2023, IRIC celebrates its 20th anniversary. Among the initiatives put in place to celebrate this anniversary, the series of portraits entitled “The Pillars of IRIC” will highlight the people who have contributed since the very beginning, sometimes in the shadows, to make IRIC what it has become.
Today, meet Sandra Weber, Research Advisor in Marc Therrien’s laboratory at IRIC.
![]()
-
November 27, 2023 The Pillars of IRIC: meet Jinny Émond
In 2023, IRIC celebrates its 20th anniversary. Among the initiatives put in place to celebrate this anniversary, the series of portraits entitled “The Pillars of IRIC” will highlight the people who have contributed since the very beginning, sometimes in the shadows, to make IRIC what it has become.
Today, meet Jinny Émond, Animal Care Worker In vivo Biology Core Facility.
![]()
-
October 31, 2023 A CRS grant to validate therapeutic targets in liver cancer
The team led by Sylvain Meloche, Director of IRIC’s Signaling and Cell Growth Research Unit, has been awarded a two-year grant of $125,000 for the project “Advanced preclinical validation of SRC-family kinases as therapeutic targets in liver cancer”. This grant is funded in partnership with the Canadian Liver Foundation.
![]()
-
October 31, 2023 Lucyle Depoërs, doctoral student and science communicator, presents the magazine L’ÉclairSci
IRIC is proud to support the creation of the new magazine l‘ÉclairSci. Initiated by doctoral student Lucyle Depoërs, from Claude Perreault’s laboratory, the project will present scientific studies carried out by IRIC teams in the form of popularized infographics. In a digital form, l‘ÉclairSci aims to share the latest advances in cancer research with the general public.
![]()
-
October 30, 2023 A new platform to predict the safety of drug compounds
With their ability to activate multiple signaling pathways at different locations in the cell, G protein-coupled receptors (GPCRs) are prime targets for drug development. Shane C. Wright, from the Karolinska Institute in Sweden, has managed to harness new technology developed during his time as a postdoctoral fellow in the laboratory of Michel Bouvier, CEO of IRIC and Director of its Molecular Pharmacology Research Unit, that can potentially be used to predict adverse effects of new treatments prior to their use in the clinic. The study, a joint effort by the Swedish and Montreal teams, has been published in the journal Nature Communications.
![]()
-
October 27, 2023 Success for the IRIC 2023 symposium!
On October 19 and 20, over 250 guests gathered for IRIC’s international scientific symposium. The event was also an opportunity for everyone to officially mark IRIC’s 20th anniversary at Université de Montréal.
![]()
-
September 27, 2023 $13,000 raised as part of the Beneva Montreal Marathon
The Beneva Montreal Marathon took place on September 23 and 24 and IRIC is proud to have collected more than $13,000 thanks to generous donors.
![]()
-
September 26, 2023 Celebration of IRIC’s 20th anniversary and its community!
On September 18, 300 people from IRIC’s student and staff communities gathered to celebrate the Institute’s 20th anniversary.
![]()
-
September 20, 2023 Announcement of the winners of the competition Science Réflexion 2023
IRIC is pleased to announce the winners of the Science Réflexion 2023 competition. This annual competition showcases the creativity and talent of the IRIC community and presents science from a different angle.
![]()
-
September 19, 2023 Congratulations to the 3 recipients of IRIC’s Salary Awards Competition for Postdoctoral Fellows
Three postdoctoral fellows won the IRIC Postdoctoral Fellowship Competition this year. These three grants were awarded thanks to dedicated donations.
![]()
-
September 7, 2023 IRIC on the road: a student recruitment tour powered by the Academic Affairs team
As part of its IRIC on the road student recruitment tour, the Institute’s academic affairs team will participate this fall in several graduate studies fairs organized by Canadian universities.
![]()
-
September 6, 2023 Pasquale Gentile: a patient treated with the UM171 molecule visits IRIC
On July 14, Pasquale Gentile, a patient who received cell expansion therapy based on the UM171 molecule, visited IRIC’s laboratories to visit the Drug Discovery Unit and meet Stéphane Gingras, the chemist who synthesized the UM171 molecule.
![]()
-
September 6, 2023 KIF18A: a promising anticancer target for combinatorial therapy with PLK1 inhibitors
The PLK1 kinase is involved at multiple levels in the control of cell division. Cancer cells are often particularly dependent on the activity of this enzyme. However, the anticancer therapies developed to target PLK1 remain largely ineffective to date. The laboratory of Professor Vincent Archambault, Director of IRIC’s Cell Cycle Regulation Research Unit, collaborated with the laboratory of Professor Michael Tyers, then Director of IRIC’s Systems Biology and Synthetic Biology Research Unit, to better understand the cellular functions of PLK1 and to identify genes whose inactivation interferes with cell proliferation in synergy with existing PLK1 inhibitors. The results suggest that inhibition of the kinesin KIF18A could be a therapeutic avenue to exploit in combination with PLK1 inhibitors. Published in the journal PLOS Genetics, the study was led by research advisor Karine Normandin.
![]()
-
August 24, 2023 Captain Mathilde: combining racing and science for the benefit of research
Research advisor in the laboratory of Sylvain Meloche, who heads the Signalling and Cell Growth Research Unit, Mathilde Soulez has agreed to assume for a third year the captain’s role for Team IRIC at the Marathon Beneva de Montréal. Scientist, runner, coach, mother; meet a woman of many talents and interests.
![]()
-
August 24, 2023 The Pillars of IRIC: meet Madeleine Héroux
In 2023, IRIC celebrates its 20th anniversary. Among the initiatives put in place to celebrate this anniversary, the series of portraits entitled “The Pillars of IRIC” will highlight the people who have contributed since the very beginning, sometimes in the shadows, to make IRIC what it has become.
Today, meet Madeleine Héroux, Biology Assistant Director, Drug Discovery Unit
![]()
-
August 18, 2023 IRIC Intern Day Conference : An opportunity to learn about scientific communication
The fifth edition of IRIC Intern Day Conference was held on Thursday, August 10. The event, organized by IRIC Academic Affairs Office together with the summer interns, enabled 11 interns to learn about scientific communication in front of their peers and IRIC colleagues.
A total of four oral presentations and seven poster presentations were given.
![]()
-
August 2, 2023 The Pillars of IRIC: meet Simon Girard
In 2023, IRIC celebrates its 20th anniversary. Among the initiatives put in place to celebrate this anniversary, the series of portraits entitled “The Pillars of IRIC” will highlight the people who have contributed since the very beginning, sometimes in the shadows, to make IRIC what it has become.
Today, meet Simon Girard, Research Officer in Guy Sauvageau’s laboratory.
![]()
-
August 1, 2023 Identification of the Emerin protein as a target of the PP2A-B55 phosphatase: a key regulation for mitosis exit
IRIC’s Cell Cycle Regulation Research Unit and Proteomics and Mass Spectrometry Research Unit, headed respectively by Professors Vincent Archambault and Pierre Thibault, have taken advantage of the fruit fly Drosophila melanogaster to identify PP2A-B55 target proteins. Their results reveal that the nuclear envelope protein Emerin is dephosphorylated by PP2A-B55, a regulation event that is important for nuclear envelope reformation and embryonic development. Conducted jointly by doctoral students Virginie Emond-Fraser and Myreille Larouche, the study is published in the Royal Society‘s Open Biology journal.
![]()
-
July 24, 2023 Four IRIC teams obtain funding from the Canadian Institutes of Health Research
The Canadian Institutes of Health Research (CIHR) recently awarded the Project grants for the Spring 2023 competition. This program aims to support original ideas with the potential to advance basic and applied knowledge in health. Across the country, 381 research projects, representing a total investment of $325 million, were funded. Four IRIC projects have received these prestigious grants.
![]()
-
July 19, 2023 UdeM Summer Immersion Program: an opportunity for the next generation to learn about molecular biology in our laboratories
On July 4, as part of the Université de Montréal Summer Immersion Program, 8 young people aged 15 to 17 took part in a thematic workshop in IRIC’s laboratories. Led by doctoral students Fadia Boudghene-Stambouli, Eloïse Duramé and Chloé Tesnière, the half-day workshop gave them the opportunity to experiment and learn about molecular biology and cancer research.
![]()
-
July 6, 2023 Identification of a novel pro-motility pathway involving the protein CNK2 in cancer cells
At IRIC, Marc Therrien and his team are interested in understanding how signal transduction is disrupted in cancer cells, allowing them to divide and spread throughout the body even in the absence of appropriate signals. Their recent work, which was led by the PhD students Jessica Gagnon and Guillaume Serwe, and the research associate David Kachaner, was published in Nature Communications and describes a new signalling pathway in which the scaffold protein CNK2 plays a central role to increase the motility of cancer cells. The research teams of Gregory Emery and Sylvain Meloche at IRIC also contributed to this study.
![]()
-
July 5, 2023 Visit the new IRIC store!
IRIC is proud to launch its new online store. Whether for a sporting event or simply on a daily basis, make the Institute shine by wearing its colors.
![]()
-
July 5, 2023 The Leukemia & Lymphoma Society of Canada presents two awards in honour of IRIC members
The Leukemia & Lymphoma Society of Canada (LLSC) presents six awards each year to recognize the contributions of the organization’s volunteers and staff. Two of these awards are named in honour of IRIC members: the Dr. Trang Hoang – Healthcare Professional of the Year Award and the William Brock – Courage to Inspire Award. IRIC is very proud to be able to count on these two models of commitment on a daily basis.
![]()
-
July 4, 2023 ENPP1 reduces the immune response in HER2+ breast cancers
HER2∆16 is an oncogenic variant of the human epidermal growth factor receptor (HER2); it is implicated in breast cancers and other types of tumors as a motor for tumorigenesis and the metastatic process. The laboratory of Professor Philippe Roux, Director of the Cell Signaling and Proteomics Research Unit at IRIC, collaborated with the team of William Muller, from the Rosalind and Morris Goodman Cancer Institute of McGill University, to characterize the molecular mechanisms underlying HER2Δ16-mediated oncogenicity. The results obtained suggest that HER2Δ16 alters the tumor microenvironment via the ENPP1 protein, which could prove to be an interesting therapeutic target. Published in the journal Cancer Immunology Research, the study was conducted jointly by Jonathan Boucher, postdoctoral fellow in the laboratory of Philippe Roux, and Sherif Samer Attalla, doctoral student in the Muller laboratory.
![]()
-
June 28, 2023 2023 Student Recruitment Event: an edition of meetings and encounters
The 2023 edition took place from June 7 to 9 and welcomed 23 participants from 19 universities around the world. More than 100 interviews were scheduled this year between participants and the 12 Institute researchers present at the event.
![]()
-
June 28, 2023 The Pillars of IRIC: meet Isabelle Caron
In 2023, IRIC celebrates its 20th anniversary. Among the initiatives put in place to celebrate this anniversary, the series of portraits entitled “The Pillars of IRIC” will highlight the people who have contributed since the very beginning, sometimes in the shadows, to make IRIC what it has become.
Today, meet Isabelle Caron, Animal Health Technician in vivo Biology Core Facility
![]()
-
June 15, 2023 Rector’s Award presented to Julie Mantovani for the Connexion Breaks project
The Rector’s Awards 2023 of the Université de Montréal were presented on June 12 to celebrate the achievements and commitment of outstanding staff members who exemplify university values. IRIC congratulates Julie Mantovani, Head of Academic Affairs, who received the Innovation Award during the ceremony. This award celebrates an individual who dares to bring change by proposing and implementing new ideas.
![]()
-
June 13, 2023 The Pillars of IRIC: meet Raphaëlle Lambert
In 2023, IRIC celebrates its 20th anniversary. Among the initiatives put in place to celebrate this anniversary, the series of portraits entitled “The Pillars of IRIC” will highlight the people who have contributed since the very beginning, sometimes in the shadows, to make IRIC what it has become.
Today, meet Raphaëlle Lambert, Manager of the Genomics Core Facility of IRIC.
![]()
-
June 2, 2023 IRIC Next Generation Awards Program 2023 scholarship recipients arrive at the Institute
This fellowship competition enables undergraduate students to spend 12 to 16 weeks at IRIC during the summer term.
This opportunity is made possible through the IRIC Scientists of Tomorrow 2023 Scholarships competition, supported financially by IRIC’s principal investigators and philanthropic funds.![]()
-
June 2, 2023 Juliette Humeau wins People’s Choice Award at Science POP provincial final
The provincial final of the Science POP science communication competition was held on May 27 and 28. In the popularization category, Juliette Humeau, a postdoctoral student in Claude Perreault’s laboratory, won the People’s Choice Award for her presentation entitled “L’action coordonnée des cellules immunitaires dans la lutte anticancéreuse” (“Coordinated action of immune cells in the fight against cancer”).
![]()
-
May 31, 2023 Philippe Roux appointed Scientific Director of IRIC
IRIC is pleased to announce the appointment of Professor Philippe Roux as Scientific Director the Institute. The Council of the Université de Montréal recently ratified the recommendation of IRIC’s Board of Directors for a two-year term. Professor Roux thus becomes the third Scientific Director in the history of the Institute, which celebrates its 20th anniversary in 2023. He succeeds to Professor Marc Therrien, who has held the position with distinction since 2013.
![]()
-
May 31, 2023 IRIC 2023 Scientific Day highlights new generation of scientists
IRIC’s 10th Scientific Day, held on May 26, showcased the work of the Institute’s scientific community. Over 150 people attended the event, which featured 12 oral presentations and 63 poster presentations. These sparked lively discussions and exchanges, which will provide food for thought for all.
![]()
-
May 30, 2023 The Pillars of IRIC: meet Caroline Baril
In 2023, IRIC celebrates its 20th anniversary. Among the initiatives put in place to celebrate this anniversary, the series of portraits entitled “The Pillars of IRIC” will highlight the people who have contributed since the very beginning, sometimes in the shadows, to make IRIC what it has become.
Today, meet Caroline Baril, Research Officer in Marc Therrien’s Laboratory at IRIC.
![]()
-
May 26, 2023 Richard and Edith Strauss Foundation: renewed support of $300,000!
Since 2019, with generous support from the Richard and Edith Strauss Foundation, researchers and their teams have been able to further investigate pediatric leukemia and acute myeloid leukemia (AML), pushing the boundaries of science on these types of cancer.
![]()
-
May 26, 2023 Renewal of the mandate of Michel Bouvier as Chief Executive Officer of IRIC
At the 0178th meeting held on April 11, 2023, the Executive Committee of the Université de Montréal appointed Michel Bouvier as Chief Executive Officer of the Institute for Research in Immunology and Cancer (IRIC), for a fourth term beginning June 1, 2023 and ending May 31, 2024.
![]()
-
May 25, 2023 A NOVA grant for Delphine Bouilly and her partners
IRIC congratulates Delphine Bouilly, Director of the Design and Application of Electronic Nanobiosensors Research Unit, who was awarded a NOVA grant of $225,000 over three years for the project “Functionalized bilayer graphene devices: achieving selectivity and sensitivity”. Offered jointly by the Fonds de recherche du Québec – Nature et technologies and the Natural Sciences and Engineering Research Council of Canada, this grant supports research projects led by up-and-coming researchers from Québec in collaboration with researchers from other Canadian provinces and territories.
![]()
-
May 17, 2023 Principal Investigator Geneviève Deblois wins Forbeck Award !
Geneviève Deblois, Director of IRIC’s Epigenetic Mechanisms and Cancer Metabolism Research Unit, is one of the 13 recipients of the 2022-2023 Forbeck Prize. “This award will allow me to be part of a group of specialized researchers, many of whom are internationally recognized in fields of research related to mine,” explains Geneviève Deblois.
![]()
-
May 17, 2023 Identification of supervillin as an ERK3 target: implications for the completion of cytokinesis
Despite potential roles in the tumorigenesis of lung and breast cancer cells, the atypical protein kinase ERK3, a member of the Ras/MAPK pathway, is poorly characterized. The laboratory of Professor Sylvain Meloche, Director of the Signalling and Cell Growth Research Unit, collaborated with the laboratory of Professor Jean-Claude Labbé, Director of the Cell Division and Differentiation Research Unit, both at IRIC, to identify ERK3 targets and better understand its cellular functions. The work carried out led to the identification of supervillin as a target of ERK3. Published in the Journal of Cellular Physiology, the study was jointly led by postdoctoral fellow Joaquim Javary and research advisor Eugénie Goupil, with the collaboration of research advisor Mathilde Soulez.
![]()
-
May 5, 2023 Thirteen IRIC members are recipients of scholarships and grants from the Fonds de recherche du Québec for the year 2023-2024
The Fonds de recherche du Québec (FRQ) offers $58.6 million in training grants, $13.1 million in career grants and $29.8 million in research grants for a total of $101.5 million in new grants for the year 2023-2024. IRIC is proud to count several of its members among the winners of these competitions.
![]()
-
May 5, 2023 Three finalists will represent IRIC at the provincial Science Pop final
Held on April 12, IRIC’s internal competition awarded three finalists who will take part in the provincial finals of the Science Pop pan-Quebec science communication contest on May 27 and 28.
![]()
-
May 4, 2023 The Pillars of IRIC: meet Éric Bonneil
In 2023, IRIC celebrates its 20th anniversary. Among the initiatives put in place to celebrate this anniversary, the series of portraits entitled “The Pillars of IRIC” will highlight the people who have contributed since the very beginning, sometimes in the shadows, to make IRIC what it has become.
Today, meet Éric Bonneil, Manager of the Proteomics Core Facility.
![]()
-
May 4, 2023 IRIC’s benefit gala raises $505,000 for Audace 2023
On April 27, over 250 guests gathered for IRIC’s (Institute for Research in Immunology and Cancer of the Université de Montréal) 20th anniversary at its benefit-event, Audace. Thanks to the generosity of guests and donors, over $505,000 were raised to sustain IRIC’s mission and improve cancer-fighting therapies.
![]()
-
May 4, 2023 The Université de Montpellier awards an honorary doctorate to Michel Bouvier
An honorary doctorate has been awarded to Michel Bouvier for his work on G protein-coupled receptors (GPCRs) and his contribution to scientific advances in the field of molecular pharmacology. The highest distinction in higher education was awarded to him by the University of Montpellier during a ceremony held on April 13.
![]()
-
May 2, 2023 Congratulations to IRIC Travel Awards - Winter 2023
The objective of the competition is to encourage IRIC M.Sc. and Ph.D. students, as well as postdoctoral fellows, to participate in major scientific congresses taking place outside the province of Quebec.
![]()
-
April 4, 2023 Discovery of new roles for eIF4E: widescale reprogramming of splicing to change the message
The team of Professor Katherine Borden, Director of the Structure and Function of the Cell Nucleus Research Unit at IRIC, set out to understand how alternative splicing was hijacked in certain patients with Acute Myeloid Leukemia (AML) who do not harbour the DNA mutations usually associated with splicing dysregulation. The study of laboratory models and samples from leukemia patients has made it possible to discover that the splicing of thousands of RNAs could be altered in an unsuspected way by the oncogenic protein eIF4E, conferring cancerous properties to the cells. Published in The EMBO Journal, the work was jointly led by postdocs Mehdi Ghram and Gavin Morris, along with research associate Biljana Culjkovic-Kraljacic.
![]()
-
March 14, 2023 More than $10,000 raised for the first FOCUS!
From February 9 to February 12, 2023 was held the first edition of FOCUS, an exhibition presenting science from an artistic perspective thanks to the creative talents of IRIC’s scientific community. The exhibition brought to light scientific works created by the Institute’s community at Projet Casa, a Montreal-based showroom founded by long-time donator : Danielle Lysaught and Paul Hamelin.
![]()
-
March 14, 2023 The Pillars of IRIC: meet Nadine Mayotte
In 2023, IRIC celebrates its 20th anniversary. Among the initiatives put in place to celebrate this anniversary, the series of portraits entitled “The Pillars of IRIC” will highlight the people who have contributed since the very beginning, sometimes in the shadows, to make IRIC what it has become.
Today, meet Nadine Mayotte, Research Advisor in Guy Sauvageau’s laboratory at IRIC.
![]()
-
March 14, 2023 Principal investigator Gregory Emery awarded a grant from the Canadian Institutes of Health Research
The Canadian Institutes of Health Research (CIHR) Project Grants support original ideas with the potential to advance basic and applied health knowledge. The most recent competition, held in the fall of 2022, funded 382 applications for a total investment of $325 million. IRIC congratulate Gregory Emery, Director of its Vesicular Transport and Cell Signalling Research Unit, who has been awarded a five-year $963,900 grant for the project “Regulation of collective cell migration by the kinase Misshapen”.
![]()
-
March 8, 2023 Principal Investigator David Knapp receives a Terry Fox Research Institute Award
David Knapp, director of IRIC’s Cellular Engineering Research Unit, is among the 2023 recipients of the Terry Fox New Investigator Awards. He will receive $433,850 over three years for the project “Understanding the Cellular Mechanisms of Age Associated Clonal Hematopoiesis”.
![]()
-
March 8, 2023 Signature of an exclusive licensing agreement and a new research collaboration in oncology with Ipsen
The Institute for Research in Immunology and Cancer of the Université de Montréal (IRIC) is proud to announce that Ipsen has exercised its option to acquire exclusive rights to one of IRIC’s pre-clinical oncology programs, which was the result of a successful research collaboration between Ipsen, IRICoR and the Université de Montréal, initiated in May 2020. Ipsen will now lead all further activities for the worldwide development and commercialization of the resulting drug candidate.
![]()
-
March 7, 2023 IRIC Master’s and Doctoral Scholarships - Fall 2022 Competition Recipients
The objective of the 2022-2023 IRIC Master and Doctoral Awards Competition is to support IRIC students who have an excellent academic and research records, but do not benefit from substantial scholarship. These M.Sc. and Ph.D. scholarships are offered thanks to the support of IRIC’s large community: its employees and researchers, its generous donors and partners.The granting of these scholarships is also made possible thanks to the involvement of several volunteers.
![]()
-
February 16, 2023 IRIC adopts new guidelines for the payment of Master’s and doctoral scholarships
In the current inflationary context, IRIC adopted in January 2023 new measures to improve the living and working conditions of its student community. With the cost of living having increased over two decades, and particularly in the last two years, the new guidelines adopted by IRIC will help reduce the financial stress of its student community and provide a healthier climate in which to conduct studies.
![]()
-
February 13, 2023 The Pillars of IRIC: meet Gabrielle Jacob
In 2023, IRIC celebrates its 20th anniversary. Among the initiatives put in place to celebrate this anniversary, the series of portraits “The Pillars of IRIC” will highlight the people who have contributed since the very beginning, sometimes in the shadows, to make IRIC what it is today. Today, meet Gabrielle Jacob, Administrative Technician in IRIC’s Finance team.
![]()
-
February 6, 2023 Renewed support for Fonds Valérie Blais
Renewed support for Fonds Valérie Blais
![]()
-
February 2, 2023 IRIC and its partners meet in Montpellier to develop an international Master’s degree program
A collaboration between IRIC, the Université de Montréal, the Université de Montpellier in France and the Università di Pavia in Italy, has led the project “MAD4Cancer – Towards an International Master Degree in Cancer Biology” to be awarded an Erasmus Mondus Design Measures grant of 55,000 euros for 15 months. The objective of the project is to set up an international Master’s degree program in cancer biology allowing students to attend the three partner universities.
![]()
-
January 27, 2023 The Fondation Jean Gaulin donates $24,420 to IRIC’s General Fund
The mission of the private family foundation Jean Gaulin is to support the well-being of the community. For many years, the foundation has supported the Institute and seek to deepen knowledge in immunology and cancer. This donation will contribute to convert the discoveries of researchers into innovative therapeutic solutions for the well-being of people affected by cancer.
![]()
-
January 20, 2023 William Brock Appointed Chair of IRIC’s Board of Directors
IRIC announces the appointment of William Brock, Lawyer Emeritus of the Québec Bar Association, as new Chairman of the Board of Directors.
![]()
-
December 7, 2022 The use of the CAXII marker could improve the classification of breast tumors
Breast cancer is a heterogeneous disease, which cannot be effectively targeted by a common, single treatment. In two-thirds of cases, the estrogen receptor alpha (ERα), a receptor activated by sex hormones, is expressed and can be targeted therapeutically by hormone therapy. However, ERα levels can vary between and within tumors, complicating its detection and subsequent choice of therapy. The progesterone receptor (PR) is commonly used clinically as a co-marker for selection of tumors qualifying for hormone therapy. Indeed, this estrogen-regulated marker is thought to reflect ERa activity. In a new study published in the journal Cancers, Sylvie Mader, director of the Molecular Targeting in Breast Cancer Treatment Research Unit at IRIC, and her PhD student Lucas Porras show that PR detection imperfectly reflects ERα activity and identify a new, more reliable co-marker.
![]()
-
November 28, 2022 A bone marrow transplant has united Dr. Claude Perreault and Dr. Robert Patenaude for 40 years now
November 2022 marks the 40th anniversary of a key encounter that allowed Robert Patenaude to become one of the first recipients of a bone marrow transplant in Canada. Here is a look back at the story that has united Robert Patenaude and Claude Perreault for four decades.
![]()
-
November 24, 2022 Identification of CDK12 as a new target of the RAS/MAPK pathway: implications for the treatment of melanoma
The laboratory of Professor Philippe P. Roux, Director of the Cell Signalling and Proteomics Research Unit, focused on the mechanisms associated with this resistance. To discover more effective anti-cancer targets, the team sought to identify the key effectors of the RAS/MAPK pathway, a signaling pathway that is overactivated in melanoma. The work carried out identified the CDK12 protein as a direct target of this pathway and highlighted its important role in melanoma cells. The project was led by postdoctoral fellow Thibault Houles and is the subject of a publication in the journal Nature Communications.
![]()
-
November 21, 2022 Brian Wilhelm and partners received over $2 million for pediatric leukemia research project
An unprecedented partnership bringing together the Conseil québécois sur la découverte du médicament (CQDM), the Canadian Cancer Society (CCS), the Cole Foundation, the Oncopole, philanthropists and Quebec companies has awarded $8,485,132 to four teams Montreal-based for their research projects on pediatric cancers. Among them is the team of Brian Wilhelm, director of the High Throughput Genomics Research Unit at IRIC and Associate Professor in the Department of Medicine of the Faculty of Medicine of the Université de Montréal.
![]()
-
October 25, 2022 Objectives met for sporting events benefiting IRIC
This fall, our community had the opportunity to join three renowned sports initiatives by participating in various competitions and/or supporting IRIC’s cause. In total, $21,325 was donated to the Institute thanks to the generosity and determination of our sports enthusiasts! A look back at these events.
![]()
-
October 24, 2022 A young researcher who has the wind in his sails
David Knapp, director of the Cellular Engineering Research Unit at IRIC and assistant professor in the Department of Pathology and Cellular Biology of the Faculty of Medicine of the Université de Montréal, has amassed successes, just two years after the establishment of his research group. As one of the most recent recipients of the Cancer Research Society’s annual grant competition, David Knapp and his team also collaborated on two studies published this summer. Here is a look back at their accomplishments.
![]()
-
October 21, 2022 Postdoctoral researcher Juliette Humeau obtains a salary grant
As part of its most recent Salary Awards Competition for Postdoctoral Fellows, held this fall, IRIC awarded a postdoctoral scholarship thanks to the Power Corporation Canada fund for Next Generation Scientists. Juliette Humeau, a postdoctoral fellow in Claude Perreault’s laboratory, was granted this funding because of the quality of her academic and scientific record.
![]()
-
October 13, 2022 The Grandes Retrouvailles at IRIC: when the doors of the laboratories open to the general public
The Université de Montréal held its first Grandes Retrouvailles on its various campuses, from September 28 to October 2. As part of the event, which included some 50 activities, 17 people were able to visit IRIC’s facilities and discover its cancer research model.
![]()
-
October 11, 2022 Major breakthrough from international collaboration paves the way for pain relief
The project is the result of an international collaboration between research groups from different fields: Brian Shoichet and Allan Basbaum, both researchers at the University of California San Francisco, Peter Gmeiner from Freidrichs Alexander University in Germany, Yang Du from the Chinese University of Hong Kong, and Michel Bouvier, IRIC’s Director General and Director of the Molecular Pharmacology Research Unit. This work is published in the journal Science.
![]()
-
October 3, 2022 Two IRIC projects selected in the Cancer Research Society’s most recent annual competition
The Cancer Research Society (CRS) has announced the recipients of 90 new research grants. Two teams from IRIC have been selected.
![]()
-
September 23, 2022 Two new recipients are awarded Power Corporation of Canada Next Generation of Scientists Postdoctoral Fellowships
The Power Corporation of Canada Next Generation of Scientists Postdoctoral Fellowships competition, held this summer, awarded funding to two IRIC postdoctoral fellows based on the quality of their research work.
![]()
-
September 16, 2022 Four IRIC students obtain funding to present their research results outside Quebec
As part of its most recent Travel Awards competition, IRIC awarded scholarships to four doctoral students. This financial support will enable them to present their research results at scientific conferences held outside Quebec.
![]()
-
September 12, 2022 The Exceptional Commitment of the Katelyn Bedard Bone Marrow Association
IRIC wishes to highlight the great generosity of the Katelyn Bedard Bone Marrow Association, which has been supporting the work of Dr. Claude Perreault’s team for 15 years. In 2022, a donation of $10,000 was made and to date, $90,000 has been donated to the Perreault laboratory to further research into hematopoietic cell transplantation and leukemia treatment.
![]()
-
September 9, 2022 IRIC on the road: the academic affairs team embarks on a recruitment tour
As part of its IRIC on the road student recruitment tour, the Institute’s academic affairs team will participate this fall in several graduate studies fairs organized by Canadian universities.
![]()
-
September 7, 2022 Katherine Borden elected to the Royal Society of Canada
Professor Katherine Borden, Director of the Structure and Function of the Cell Nucleus Research Unit at IRIC and Full Professor in the Department of Pathology and Cell Biology in the Faculty of Medicine at the Université de Montréal, has been elected as a Fellow of the Academy of Science of the Royal Society of Canada (RSC).
![]()
-
August 22, 2022 A chemical molecule designed at IRIC has been named “Molecule of the Month” in June 2022 by the Drug Hunter platform
Clinical candidate designed and synthesized at IRIC by the Drug Discovery Unit led by Principal Investigator Anne Marinier, the molecule BMS-986120 (UDM-002985) has been named “Molecule of the Month” by the Drug Hunter Platform.
![]()
-
August 11, 2022 IRIC Intern Day Conference: a great experience to learn how to communicate research results
The fourth edition of the IRIC Intern Day Conference was held on Thursday, August 11, 2022. The event, organized by the IRIC Academic Affairs Office together with the summer interns, enabled 10 interns to learn about scientific communication.
![]()
-
July 21, 2022 Discovery of a new therapeutic avenue targeting the cell cycle in a subtype of acute myeloid leukemia
The project, led by research officer Céline Moison, was published in the journal Blood Advances. The work was carried out in collaboration with the teams of Guy Sauvageau and Anne Marinier at IRIC, Josée Hébert at Hôpital Maisonneuve-Rosemont and Vincent-Philippe Lavallée at CHU Sainte-Justine.
![]()
-
July 19, 2022 Four IRIC researchers obtain funding from the Canadian Institutes of Health Research
Resulting from the most recent research grant competition of the Canadian Institutes of Health Research (CIHR), four IRIC researchers obtained funds via the Project grant program.
![]()
-
July 8, 2022 Five IRIC graduates named to the Honor List of the Rector of the Université de Montréal
IRIC is proud to count five of its graduates among the most recent appointments to the Rector’s Honor List.
![]()
-
July 5, 2022 Three IRIC researchers receive funding from the Natural Sciences and Engineering Research Council of Canada
The Natural Sciences and Engineering Research Council of Canada (NSERC) recently announced the results of its 2022 Research Grant Competition. More than $430 million will be awarded to researchers across the country to their research programs well. Three IRIC investigators were awarded grants.
![]()
-
June 29, 2022 IRIC recruits Dr. Vincent Q. Trinh to be a Principal Investigator
The Institute for Research in Immunology and Cancer (IRIC) of the Université de Montréal is proud to welcome aboard a new Principal Investigator in the Digital Histology and Advanced Pathology Research Unit, Dr. Vincent Q. Trinh, who will take up his position on July 1st, 2022.
![]()
-
June 29, 2022 Meet Vincent Q. Trinh, new Principal Investigator at IRIC
The Institute for Research in Immunology and Cancer (IRIC) of the Université de Montréal is proud to welcome aboard a new Principal Investigator in the Digital Histology and Advanced Pathology Research Unit, Dr. Vincent Q. Trinh, who will take up his position on July 1st, 2022. Dr. Trinh enthusiastically agreed to answer a few questions.
![]()
-
June 27, 2022 Hyperactivation of the NOTCH1 pathway in T-cell acute lymphoblastic leukemia: two recent publications bring new insights
The team of Trang Hoang, director of the Hematopoiesis and Leukemia Research Unit at IRIC, focused on the hyperactivation of NOTCH1 in T-ALL. The work of Diogo Veiga, postdoctoral fellow now professor of Genomics and Bioinformatics at the Universidade Estadual de Campinas in Brazil, and Mathieu Tremblay, research advisor, has led to the publication of two articles.
![]()
-
June 22, 2022 Michel Bouvier appointed Knight of the Ordre national du Québec
On June 22, 2022, Michel Bouvier, Chief Executive Officer of IRIC, Principal Investigator of the Molecular Pharmacology Research Unit and Professor in the Department of Biochemistry and Molecular Medicine of the Faculty of Medicine of the Université de Montréal, is appointed Knight of the Ordre national du Québec.
![]()
-
June 21, 2022 2022 IRIC Next Generation Awards : twelve interns will train at IRIC this summer
Twelve students have the opportunity to complete a research internship for 12 or 16 weeks at IRIC, and to receive a scholarship of excellence of respectively $4,800 or $6,400.
![]()
-
June 21, 2022 A 2022 edition in person for the IRIC Student Recruitment Event
The 2022 edition took place from June 1 to 3 and welcomed 29 participants from 18 universities around the world. 153 interviews were scheduled this year between participants and the 14 Institute researchers present at the event.
![]()
-
June 8, 2022 Nine next generation scientists from IRIC receive a training award from the Fonds de recherche du Québec
The Fonds de recherche du Québec (FRQ) awarded $39.3 million in training grants for its 2022-2023 competitions. IRIC is proud to count nine scientists from its next generation among the recipients of these competitions.
![]()
-
May 26, 2022 Claude Perreault and Pierre Thibault, founders of Epitopea, a company that offers hope for the treatment of cancer
Founded in 2021, the transatlantic biotechnology company Epitopea is the result of collaborations between the teams of Claude Perreault and Pierre Thibault, both principal investigators at IRIC. The mission of Epitopea is to develop immunotherapies to treat different types of cancers by targeting a new class of antigens.
![]()
-
May 20, 2022 3 million in support from the Stem Cell Network to study new ways to use blood stem cells in the treatment of leukemia
Guy Sauvageau, principal investigator at IRIC, hematologist and full professor in the Department of Medicine of the Faculty of Medicine of the Université de Montréal, is the recipient of major funding worth $3 million over three years. The project led by Dr. Sauvageau is composed of eight research groups, including those of Philippe Roux and Étienne Gagnon, both researchers at IRIC.
![]()
-
May 16, 2022 Vincent Archambault appointed Full Professor in the Department of Biochemistry and Molecular Medicine at the Université de Montréal
IRIC is proud to announce the promotion of Vincent Archambault, Principal Investigator and Director of the Cell Cycle Regulation Research Unit, as Full Professor in the Department of Biochemistry and Molecular Medicine at the Université de Montréal.
![]()
-
May 12, 2022 Les Rendez-vous Audace 2022: Dr. Eralda Kina answers your questions
Les Rendez-vous Audace of the IRIC, held on April 27, brought together André Robitaille and three inspiring speakers around the theme “A world in transformation, a next generation in action”. Eralda Kina, oncologist and doctoral student at IRIC in the laboratory of Claude Perreault, discussed her experience as a clinician and the great hopes she has in immunology and cancer research. Dr. Kina generously agreed to answer the questions received from the public during the broadcast of Les Rendez-vous Audace.
![]()
-
May 9, 2022 Geneviève Deblois Honoured by the Canadian Institutes of Health Research
Geneviève Deblois, Principal Investigator at IRIC and Assistant Professor at the Faculty of Pharmacy of the Université de Montréal, is the recipient of the Early Career Investigator in Cancer Award 2022. This award, presented by the Institute of Cancer Research of the Canadian Institutes of Health Research (CIHR), recognizes excellence in research conducted in Canada. It is awarded to the top-ranked new investigators in the CIHR fall competition, Project Scheme.
![]()
-
April 28, 2022 Identification of a new key role for interphase microtubules at mitosis entry
At the beginning of cell division, cells round-up through different mechanisms, notably the reorganization of their cortex. Among the proteins involved in this cell morphogenesis, there is the family of ERM proteins (ezrin, radixin and moesin). The team of Professor Sébastien Carréno, Director of the Cellular Mechanisms of Morphogenesis during Mitosis and Cell Motility Research Unit, wanted to identify the molecular cue activating these proteins at mitotic entry. Their work has shed light on a novel mechanism involving interphase microtubules in the regulation of cell rounding at mitotic onset. The project, led by PhD student Kévin Leguay, is the subject of a publication in the Journal of Cell Biology.
![]()
-
April 20, 2022 New agreement for an international research collaboration in drug discovery
IRIC is pleased to announce the signing of a collaboration agreement between Ono Pharmaceutical Co, Ltd., one of the largest clinical stage pharmaceutical companies in Japan, Domain Therapeutics S.A. and the Université de Montréal to discover novel small molecules targeting G protein-coupled receptors (GPCRs) for the treatment of metabolic diseases.
![]()
-
April 12, 2022 Publication of two studies characterizing the selectivity of G protein-coupled receptors’ coupling
Two-thirds of human hormones and one-third of clinical drugs target and activates the G protein-coupled receptors. For a given receptor, different ligands can selectively engage different subsets of signaling pathways. Mapping the effectors that can be engaged by a given receptor is essential for understanding physiological processes as well as for designing safer drugs. The team of Professor Michel Bouvier, Director of the Molecular Pharmacology Research Unit and Chief Executive Officer of IRIC, worked on characterizing the coupling selectivity of GPCRs.
![]()
-
March 7, 2022 IRIC celebrates International Women’s Day
IRIC is taking advantage of International Women’s Rights Day to recognize and highlight the exceptional contribution of all its scientists who work to make a difference.
![]()
-
February 23, 2022 Assya Trofimov, Ph.D. student at IRIC, is the recipient of the prestigious Mahan postdoctoral fellowship.
Assya Trofimov, a Ph.D. student being co-directed in the laboratories headed by Dr. Claude Perreault and by Sébastien Lemieux, was awarded the prestigious Mahan postdoctoral fellowship. The competition, held annually by the Fred Hutchison Cancer Research Center (Fred Hutch) in Seattle, awards outstanding candidates by providing a 21-month stipend to carry out a research project in one of its Computational Biology laboratories.
![]()
-
February 17, 2022 Three sporting events for the benefit of IRIC
In 2022, IRIC community will have the opportunity to participate in three renowned sporting events: Les Défis du Parc national de la Mauricie, The Grands Prix Cyclistes de Québec et de Montréal, and the Marathon Beneva de Montréal.
![]()
-
February 11, 2022 IRIC marks the 2022 International Day of Women and Girls in Science
On this symbolic day, IRIC is proud to mark the exceptional contribution to cancer research made by all of its women members.
![]()
-
February 4, 2022 Four IRIC researchers receive Canadian Institutes of Health Research grants
In the Fall 2021 competition, four IRIC research teams were awarded prestigious grants.
![]()
-
February 4, 2022 The life lesson of marathoner Jean Mireault
On this World Cancer Day, IRIC would like to point out the generous contribution to our cause made by Mr. Jean Mireault. On October 11, 2021, Mr. Mireault ran in the Boston Marathon in honor of his mother, who died of cancer at a very young age. Although his race did not unfold as he had hoped because of an unexpected issue at the midway mark, Mr. Mireault drew a very inspiring lesson from the experience for all of us. Speaking very frankly, he describes the state of mind that he was in when he managed to complete, not without struggling, the second part of his marathon.
![]()
-
January 28, 2022 Renewal of Michel Bouvier’s mandate as Chief Executive Officer of IRIC
Following a meeting of the Executive Committee, held on December 7, the Université de Montréal announced the continuation of Michel Bouvier’s mandate as Chief Executive Officer of IRIC for a third one-year term.
![]()
-
January 27, 2022 IRIC obtains major funding to develop an intelligent chemolibrary
As part of the initial phase of Médicament Québec, IRIC recently obtained major funding of $3,200,045 for the Expanded Capacity and Quality of Chemolibraries for Drug Discovery Project.
![]()
-
January 26, 2022 Katherine Borden awarded by the Canadian Society for Molecular Biosciences
The Canadian Society for Molecular Biosciences (CSBM) awarded the prestigious 2022 Canadian Science Publishing Senior Investigator Award to Katherine Borden, Principal Investigator at IRIC and Full Professor in the Department of Pathology and Cell Biology at the Faculty of Medicine of the Université de Montréal.
![]()
-
January 20, 2022 Significant support from the National Institutes of Health to study new ways to target Acute Myeloid Leukemia
Professor Katherine Borden, Principal Investigator leading the Structure and Function of the Cell Nucleus Research Unit at IRIC, received a grant worth US$1.47 million (C$1.85 million) over five years. This funding from the National Institutes of Health (NIH), the largest biomedical research granting agency in the United States, will support her research project aimed at elucidating the molecular mechanisms of eIF4E-mediated tumor transformation.
![]()
-
January 18, 2022 Identification of a new oncogenic signaling pathway in the development of hepatocellular carcinoma
Hepatocellular carcinoma (HCC) is the sixth most common cancer and the third deadliest cancer in the world. Despite these impressive numbers, the few treatments currently available have only modest effects in affected patients, who are often diagnosed at a late stage of the disease. A better mechanistic understanding of HCC development is required to develop novel targeted therapies that work more efficiently. The team of Professor Sylvain Meloche, Director of the Cell Signaling and Cell Growth Research Unit at IRIC, recently shed light on a previously unknown oncogenic signaling pathway for HCC. Jean-Philippe Guégan and Marjorie Lapouge are the co-first authors of the article published in January in the journal Science Signaling.
![]()
-
December 14, 2021 IRIC Master’s and Ph.D. Scholarships – Recipients of the Fall 2021 Competition
As part of the IRIC Master’s and Ph.D. Scholarships Competition held this fall, the Institute is proud to announce the 14 recipients of this edition.
The objective of the competition is to support IRIC students with an outstanding academic and research record, but who do not benefit from any other substantial scholarships.
Congratulations to all the recipients!
![]()
-
December 13, 2021 IRIC, a key player in the Acuité-Québec project: $27M to accelerate the discovery of new drugs
The Institute for Research in Immunology and Cancer (IRIC) of the Université de Montréal is delighted to be part of the Acuité-Québec project, an initiative aimed at fostering the discovery of new drugs.
![]()
-
November 16, 2021 Recruitment of IRIC’s Scientific Director
The IRIC invites applications for the position of Scientific Director and Principal Investigator at the Institute. IRIC seeks an established scientist with an exceptional track-record who will oversee the scientific development of the institute while leading his/her own basic and/or translational research program in cancer-relevant areas.
![]()
-
November 15, 2021 Organize a fundraising activity to support research in cancerology
On this National Philanthropy Day, IRIC is giving access to a new online tool for anyone wishing to organize a fundraising activity to support the Institute.
![]()
-
September 30, 2021 Trang Hoang receives the prix Mentor scientifique 2021 from the Club de Recherches Cliniques du Québec
Trang Hoang, Principal Investigator at the Institute for Research in Immunology and Cancer (IRIC) and Full Professor in the Department of Pharmacology of the Faculty of Medicine of the Université de Montréal, has been awarded the prix Mentor scientifique 2021 (2021 Mentor Award) by the Club de Recherches Cliniques du Québec (CRCQ).
![]()
-
September 21, 2021 A forum for Gender Equality Week in Canada
As an institution, IRIC seeks to promote equity and increase diversity and inclusion, in all of its forms, throughout its programs.
![]()
-
July 29, 2021 Pierre Thibault’s team shares its knowledge on mass spectrometry-based proteomic strategies used to study UBL like modifications
The group of Pierre Thibault, Principal Investigator at IRIC and professor in the Department of Chemistry of UdeM, with a colleague from Leiden University Medical Center in the Netherlands, recently published an exhaustive review article in Nature Reviews Methods Primers to educate researchers of all levels on the variety of mass spectrometry-based proteomic strategies used to study ubiquitin-like (UBL) modifications and the type of information that can be generated through these approaches.
![]()
-
June 21, 2021 Brian Wilhelm awarded with major funding through the new Genomics Integration Program – Human Health initiated by Génome Québec
Brian Wilhelm, Principal Investigator at IRIC and Associate Professor in the Department of Medicine at the Faculty of Medicine of the Université de Montréal, is the recipient of major funding through the new Genomics Integration Program – Human Health, initiated by Génome Québec in collaboration with the Oncopole.
![]()
-
June 17, 2021 IRIC salutes the Quebec government for its commitment: $13M invested to bring together the players in the drug discovery and production field
IRIC is delighted by the creation of “Médicament Québec”, a major strategic hub whose mission is to promote greater autonomy and increase Quebec’s drive with respect to drug discovery, development and production.
![]()
-
June 3, 2021 Appointment of Clarissa Desjardins and renewal of Robert Paré and Jacques Bernier’s mandate on the IRIC’s Board of Directors
IRIC is pleased to announce the appointment of Dr. Clarissa Desjardins as a new member of the Board of Directors as well as the renewal of Mtre Robert Paré and Mr. Jacques Bernier’s mandate as a member of the Board by the Executive Committee of the Université de Montréal.
![]()
-
May 26, 2021 IRIC participates in the next Forum of the Chamber of Commerce of Metropolitan Montreal
On Monday, May 31, IRIC will take part in the Strategic Forum Universities, CEGEPs, businesses: Working together for a strong, innovative and socially responsible economy, an initiative co-developed by the Chamber of Commerce of Metropolitan Montreal and the FRQs.
![]()
-
May 17, 2021 Trang Hoang is the recipient of the 2021 CSMB Jeanne Manery Fisher Award
Trang Hoang, Principal Investigator at the Institute for Research in Immunology and Cancer (IRIC) and Full Professor in the Department of Pharmacology at the Faculty of Medicine of the Université de Montréal is the recipient of the 2021 Canadian Society for Molecular Biosciences (CSMB) Jeanne Manery Fisher Award.
![]()
-
May 14, 2021 Les Rendez-vous Audace 2021: $320,000 raised to support IRIC’s cancer research efforts
Exceptionally this year, the Institute for Research in Immunology and Cancer (IRIC) of the Université de Montréal presented a virtual formula of its traditional Audacious fundraising event: Les Rendez-vous Audace. For the occasion, approximately 615 people from 8 countries attended the three free conferences and close to 20 sponsors supported the event. Thanks to the generosity of our partners and donors, a total of $320,000 was raised to accelerate the discovery of better therapies for the benefit of patients.
![]()
-
May 11, 2021 Lecture by Dr. Roger D. Kornberg – Nobel Prize in Chemistry
On May 20th, IRIC and the Université de Montréal will have the honour of welcoming Dr. Roger D. Kornberg, Nobel Prize in Chemistry in 2006. He will present a lecture titled “Chromosome Structure and Transcription”.
![]()
-
April 26, 2021 Genome-wide screens identify a novel gene that tapers the DNA damage response in human cells with eroded telomeres
Telomeres are DNA sequences that serve as critical caps to protect the ends of chromosomes against degradation, fraying, and fusion with other chromosomes. They play an important role in maintaining genome integrity but in most cells in our body, telomeres become shorter with each cell division as the activity of the telomere maintenance enzyme (TERT) is repressed. After sufficient telomere shortening, the protective telomere structure is lost, activating the tumor-suppressor gene p53 and the DNA damage response. This leads to a non-proliferative state in normal cells called senescence, or to cell death in cancer cells. These inhibitory effects upon cell proliferation are thought to be an important mechanism to prevent the proliferation of damaged cells and the formation of tumors.
![]()
-
April 26, 2021 A pathogenic yeast has many tricks up its sleeve
Infections with the yeast Candida albicans are a significant public health problem and a common complication in immunocompromised individuals such as people with AIDS, chemotherapy patients, and organ transplant patients. Although there are some treatments, their effectiveness can be compromised by the emergence of resistant strains and the identification of new antifungals is highly desirable. A few years ago, an IRIC team identified nicotinamide (NAM), a form of vitamin B3, as a potential antifungal treatment.
![]()
-
April 21, 2021 Talk from Guy Sauvageau at the CCMM
Leveraging innovation and collaboration to accelerate scientific discoveries in oncology
![]()
-
April 13, 2021 Spatiotemporal coordination: a key aspect allowing cells to divide correctly
Enzymes that regulate cell division must be subjected to spatiotemporal control, allowing them to access the cellular structures they control only at the appropriate time. The segregation of chromosomes during mitosis is the most crucial event of cell division. Spatiotemporal coordination is therefore fundamental to the good progress of mitosis but it remains poorly understood.
![]()
-
April 1, 2021 TMEM16F: a protein with a key role in the regulation of T cell activity
By Fatéma Dodat
Understanding the regulation of T cell activity is crucial for the development of immunity against infections, but also in cancer. A study conducted by the team of Etienne Gagnon reveals that TMEM16F, a molecule that promotes the exchange of components between the lipid bilayer of a cell membrane, is involved in this activation.
![]()
-
March 30, 2021 One step closer to developing a cancer vaccine
Claude Perreault and his team published an important article related to the development of a therapeutic cancer vaccine for a type of blood cancer.
![]()
-
February 26, 2021 A new way beckons to treat severe obesity
By Mathieu-Robert Sauvé, UdeM Nouvelles
Michel Bouvier, unveils research that could someday lead to treatment for early morbid familial obesity.
![]()
-
February 17, 2021 Michel Bouvier, CEO of IRIC, takes part in the Pan-Canadian Showcase on the future of drug discovery and production in Canada
The Institute for Research in Immunology and Cancer (IRIC) of the Université de Montréal is proud to announce that Michel Bouvier, CEO of the Institute and recent recipient of the Killam Prize in Health Sciences, is taking part in the virtual event “Building Biotech: Science and Talent Accelerating Biomedical Innovation, a Pan-Canadian Showcase and Dialogue”.The Showcase is being held today, February 17, 2021, and is organized by PRiME at the University of Toronto.
![]()
-
February 4, 2021 2021 World Cancer Day: Help us vanquish cancer!
February 4 is World Cancer Day. Every year, it’s a day that is celebrated all over the world to highlight the incredible advances in cancer research that help save more and more lives and create undeniable hope. In memory of those we love who have left us too soon or in honour of those who are currently fighting the disease, let’s all highlight this Day, the progress that we have made and the progress left to be made.
![]()
-
February 3, 2021 Michel Bouvier is the laureate of the 2021 Killam Prize in Health Sciences
Michel Bouvier, CEO of IRIC and Professor in the Department of Biochemistry and Molecular Medicine of the Faculty of Medicine of the Université de Montréal, is the laureate of the 2021 Killam Prize in Health Sciences.
![]()
-
January 26, 2021 IRIC Master’s and Ph.D. Scholarships – Recipients of the Fall 2020 Competition
As part of the IRIC Master’s and Ph.D. Scholarships Competition held this fall, the Institute is proud to announce the 17 recipients of this edition.
![]()
-
January 12, 2021 UM171: the molecule that keeps on giving
In a study published in Cell Stem Cell, a research team of 15 scientists led by UdeM hematologist Guy Sauvageau demonstrates the cancer-fighting molecule’s mechanism of action.
![]()
-
December 2, 2020 Nanotechnologies: the Bouilly lab reveals important principles in the design of biocaptors
In the past decades, advances in the field of nanotechnology, especially through the discovery and production of new nanomaterials, have catalyzed remarkable innovation in the creation of different biosensors, which are analytical devices used to detect and study the properties of many biological molecules. There are many types of biosensors and it is often difficult to compare their performance and identify the most promising approaches.
![]()
-
November 9, 2020 Sébastien Lemieux and his team’s project is a recipient of the ODAC Competition
Génome Québec, IVADO and Oncopole have come together to organize the “Omics Data Against Cancer” (ODAC) Competition. This initiative aims to support projects developing applications and artificial intelligence tools used to better exploit the datasets generated by genomics and the study of cancer.
The project of Sébastien Lemieux, Principal Investigator at IRIC and his team is one of the 5 recipient projects of the program.
![]()
-
October 20, 2020 IRIC’s Academic Affairs team receives the prix d’excellence en enseignement
IRIC is proud to announce that the prix d’excellence en enseignement Catégorie Innovation – Soutien à la réussite étudiante has been awarded to its Academic Affairs team: Sébastien Carréno, Julie Mantovani, Evelyne Muhire, Pascale Le Thérizien. This recognition bestowed by the Vice-Rector of Student and Academic Affairs of the Université de Montréal underscores the outstanding contribution made by the team to student success.
![]()
-
October 15, 2020 2020 student recruitment tour: a virtual edition this fall
Each fall, the IRIC’s Academic Affairs team launches an “IRIC on the road” student recruitment tour. This year, in the exceptional context caused by the COVID-19 pandemic, our representatives are going to meet students virtually. They will participate in several graduate studies fairs organized on the web by various Canadian universities.
![]()
-
October 14, 2020 A donation benefiting IRIC and the Department of Biochemistry and Molecular Medicine of the UdeM
As part of a major planned donations campaign, led by the Réseau des diplômés et des donateurs de l’Université de Montréal, Michel Bouvier, Chief Executive Officer of IRIC and his partner, Muriel Aubry, announced their testamentary donation benefiting IRIC and the Department of Biochemistry and Molecular Medicine of the UdeM, in support of cancer research and the training of the next generation of scientists.
![]()
-
October 13, 2020 Breaking the RNA capping paradigm
This discovery by a team of IRIC researchers could soon be taught in molecular biology courses. Katherine Borden’s laboratory recently published a study in the journal PNAS that demonstrated that the activity of RNAs can be regulated in a new way: by modulating a RNA modification known as capping.
![]()
-
September 4, 2020 Three IRIC researchers receive substantial support from the Canada Foundation for Innovation
On August 18, 2020, the Canada Foundation for Innovation (CFI) announced the selected research projects that will benefit from its financial support. This substantial support allows researchers to acquire highly specialized avant-garde equipment and infrastructure, and to recruit and retain internationally renowned scientists.
![]()
-
August 6, 2020 Genome-wide screens reveal that resveratrol induces replicative stress in human cells
Resveratrol (RSV) is a natural compound found in many plants, including the skin of grapes (and in wine), blueberries, raspberries, mulberries and peanuts. It has attracted considerable research attention due to its reported beneficial properties in experimental models such as yeasts, nematode worms, fruit flies and mice, including lifespan extension under certain conditions. A causal role of RSV in human health has not yet been established.
![]()
-
April 22, 2026 Coup de cœur- April 2026
Virginie is fearless. She handles adversity with ease and stays calm to find solutions to any problems that arise. With her formidable efficiency, she manages her many projects with a masterful hand. Empathetic by nature, she takes an interest in others and doesn’t hesitate to take the time needed to ensure everyone is on the same page.
-
February 26, 2026 IRIC celebrates International Day of Women and Girls in Science!
On February 21, IRIC was present at the Montreal Science Centre for Women and Girls in Science Day. This event provides an opportunity to meet passionate women working in a wide range of fields, from technology to the environment, health to aerospace.
-
June 26, 2025 UM171 breaks new ground
Just a few years ago, there was little hope for people suffering from hematological malignancies requiring stem cell transplants, such as acute leukemia and myelodysplastic syndromes. Today, thanks to the work of Guy Sauvageau and Anne Marinier, new treatment options are gradually entering the market worldwide. Experts in the molecular genetics of stem cells and medicinal chemistry respectively, the two IRIC scientists and UdeM professors were behind the discovery of the UM171 molecule, which saw the light of day in 2014.
-
September 29, 2022 Young people set up a fundraiser for IRIC
IRIC recognizes the act of generosity of the Collège Nouvelles Frontières. During one of their “paid civilian days” where students are allowed to trade in their uniforms for the clothes of their choice, they also take advantage of this anticipated day to raise funds to support the community.
-
May 13, 2022 SQSV 2022-2025: Encouraging news for the industry!
IRIC is pleased with the announcement of the Quebec Minister of Economy and Innovation, Mr. Pierre Fitzgibbon, regarding the unveiling of the Quebec Life Sciences Strategy 2022-2025 during the renowned event Effervescence. Under the theme “Using our ingenuity to advance health”, the strategy, updated in the context of COVID-19, proposes concrete and innovative actions to promote this key industry of the Quebec economy and encourage its growth.
-
January 24, 2022 Investing in research: a move that pays off
A possible therapeutic ovarian cancer vaccine, a new treatment to fight leukemia and a molecule that could even short-circuit up to 70% of all types of cancers… These three projects, currently under development at IRIC, benefited from funds provided through private donations. Among them, publicly traded securities are a beneficial way to contribute to the next generation of cancer treatments.
-
October 4, 2021 -
September 22, 2021 2021 student recruitment tour: again, a virtual edition this fall
Each fall, the IRIC’s Academic Affairs team launches an “IRIC on the road” student recruitment tour. This year, our representatives are going to meet students virtually. They will participate in several graduate studies fairs organized on the web by various Canadian universities.